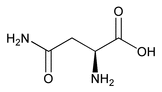
Asparagine
C4H8N2O3·H2O 150.13
C4H8N2O3 132.12
l-Asparagine;
l- -Aminosuccinamic acid, monohydrate
-Aminosuccinamic acid, monohydrate


 [5794-13-8].
[5794-13-8].
Anhydrous

 [70-47-3].
[70-47-3].
(as par' a jeen).
C4H8N2O3·H2O 150.13
C4H8N2O3 132.12
l-Asparagine;
l-
Anhydrous
DEFINITION
Asparagine is anhydrous, or contains one molecule of water of hydration. It contains NLT 98.0% and NMT 101.5% of C4H8N2O3, as l-asparagine, calculated on the dried basis.
IDENTIFICATION
• A. Infrared Absorption  197K
197K
[Note—Use USP Asparagine Anhydrous RS and USP Asparagine Monohydrate RS for the evaluation of the anhydrous and monohydrate forms of Asparagine, respectively. ]
ASSAY
• Procedure
Sample:
130 mg
Titrimetric system
(See Titrimetry  541
541 .)
.)
Mode:
Direct titration
Titrant:
0.1 N perchloric acid VS
Blank:
3 mL of formic acid in 50 mL of glacial acetic acid
Endpoint detection:
Potentiometrically
Analysis:
Dissolve the Sample with 3 mL of formic acid in 50 mL of glacial acetic acid. Perform a blank determination.
Calculate the percentage of asparagine (C4H8N2O3) in the Sample taken:
[(V  B) × N × F × 100]/W
B) × N × F × 100]/W
| V | = | = volume of Titrant consumed by the Sample (mL) |
| B | = | = volume of Titrant consumed by the Blank (mL) |
| N | = | = actual normality of the Titrant (mEq/mL) |
| F | = | = equivalency factor for asparagine, 132.1 mg/mEq |
| W | = | = weight of the Sample (mg) |
Acceptance criteria:
98.0%–101.5% on the dried basis
IMPURITIES
• Lead  251
251
Sample:
1 g
Control:
5 mL of Diluted Standard Lead Solution (5 µg of Pb)
Acceptance criteria:
NMT 5 ppm
• Chromatographic Purity
Standard solution:
0.05 mg/mL of USP Asparagine Anhydrous RS or USP Asparagine Monohydrate RS
[Note—Use USP Asparagine Anhydrous RS and USP Asparagine Monohydrate RS for the evaluation of the anhydrous and monohydrate forms of Asparagine, respectively. ]
Sample solution:
10 mg/mL
Chromatographic system
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture
Application volume:
5 µL
Developing solvent system:
Butyl alcohol, glacial acetic acid, and water (3:1:1)
Spray reagent:
2 mg/mL of ninhydrin in butyl alcohol and glacial acetic acid (19:1)
Analysis
Samples:
Standard solution and Sample solution
Proceed as directed in the general test chapter, and then dry the plate at 80 for 30 min. Spray the plate with the Spray reagent, heat at 80
for 30 min. Spray the plate with the Spray reagent, heat at 80 for 10 min, and examine under white light.
for 10 min, and examine under white light.
Acceptance criteria:
No secondary spot from the Sample solution is larger or more intense than the principal spot from the Standard solution (0.5%), and NMT 1.0% of total impurities is found.
SPECIFIC TESTS
• Optical Rotation, Specific Rotation  781S
781S
Sample solution:
10 mg/mL, in 6 N hydrochloric acid
Acceptance criteria:
+33.0 to +36.5
to +36.5 , measured at 20
, measured at 20
• Microbial Enumeration Tests  61
61 and Tests for Specified Microorganisms
and Tests for Specified Microorganisms  62
62 :
The total aerobic microbial count does not exceed 1000 cfu/g, and the total combined molds and yeasts count does not exceed 100 cfu/g.
:
The total aerobic microbial count does not exceed 1000 cfu/g, and the total combined molds and yeasts count does not exceed 100 cfu/g.
• Loss on Drying  731
731 :
Dry a sample at 130
:
Dry a sample at 130 for 3 h: the anhydrous form loses NMT 1.0% of its weight, and the monohydrate loses between 11.5% and 12.5% of its weight.
for 3 h: the anhydrous form loses NMT 1.0% of its weight, and the monohydrate loses between 11.5% and 12.5% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well-closed, light-resistant containers. Store at room temperature.
• Labeling:
Label it to indicate whether it is anhydrous or the monohydrate.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1702
Pharmacopeial Forum: Volume No. 31(1) Page 87