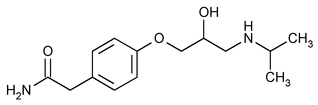
Atenolol
C14H22N2O3 266.34
Benzeneacetamide, 4-[2-hydroxy-3-[(1-methylethyl)amino]propoxy]-;
2-[p-[2-Hydroxy-3-(isopropylamino)propoxy]-phenyl]acetamide

 [29122-68-7].
[29122-68-7].
(a ten' oh lol).
C14H22N2O3 266.34
Benzeneacetamide, 4-[2-hydroxy-3-[(1-methylethyl)amino]propoxy]-;
2-[p-[2-Hydroxy-3-(isopropylamino)propoxy]-phenyl]acetamide
DEFINITION
Atenolol contains NLT 98.0% and NMT 102.0% of C14H22N2O3, calculated on the dried basis.
IDENTIFICATION
• B. Ultraviolet Absorption  197U
197U
Sample solution:
20 µg/mL in methanol
ASSAY
• Procedure
Mobile phase:
1.1 g of sodium 1-heptanesulfonate and 0.71 g of anhydrous dibasic sodium phosphate in 700 mL of water. Add 2 mL of dibutylamine, and adjust with 0.8 M phosphoric acid to a pH of 3.0. Add 300 mL of methanol, mix, and pass through a filter having a 0.5-µm or finer porosity. Degas this solution before use.
Standard solution:
0.01 mg/mL of USP Atenolol RS in Mobile phase
Sample solution:
0.01 mg/mL of Atenolol in Mobile phase. Sonicate for 5 min for complete dissolution.
Chromatographic system
Mode:
LC
Detector:
UV 226 nm
Column:
3.9-mm × 30-cm; packing L1
Flow rate:
0.6 mL/min
Injection size:
10 µL
System suitability
Sample:
Standard solution
Suitability requirements
Column efficiency:
NLT 5000 theoretical plates
Tailing factor:
NMT 2.0
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C14H22N2O3 in the portion of Atenolol taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Atenolol RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Atenolol in the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0% on the dried basis
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.2%
:
NMT 0.2%
• Chloride and Sulfate, Chloride  221
221
Sample solution:
Dissolve 1.0-g in 100 mL of 0.15 N nitric acid.
Acceptance criteria:
Shows no more turbidity with 1 mL of silver nitrate TS than 1.4 mL of 0.020 N hydrochloric acid in 100 mL of 0.15 N nitric acid (0.1%)
Organic Impurities
• Procedure
Mobile phase:
Prepare as directed in the Assay.
Sample solution 1:
0.1 mg/mL of Atenolol in Mobile phase
Sample solution 2:
0.5 µg/mL of Atenolol, from Sample solution 1 in Mobile phase
Chromatographic system:
Proceed as directed in the Assay, except use the injection size listed below.
Injection size:
50 µL
Analysis
Samples:
Sample solution 1 and Sample solution 2
[Note—Chromatograph Sample solution 1 for a period of time that is 6 times the retention time of the atenolol peak. ]
Calculate the percentage of each impurity in Sample solution 1:
Result = 0.5(rU/rA)
| rU | = | = peak response of any individual impurity in Sample solution 1 |
| rA | = | = peak response of Atenolol in Sample solution 2 |
Acceptance criteria
Individual impurities:
NMT 0.25%
Total impurities:
NMT 0.5%
SPECIFIC TESTS
• Melting Range or Temperature, Class I  741
741 :
152
:
152 –156.5
–156.5
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 to constant weight: it loses NMT 0.5% of its weight.
to constant weight: it loses NMT 0.5% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well-closed containers. Store at room temperature.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Sujatha Ramakrishna, Ph.D.
Senior Scientific Liaison 1-301-816-8349 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2260
Pharmacopeial Forum: Volume No. 35(3) Page 545