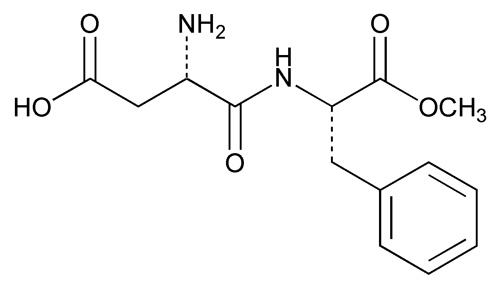
Aspartame
C14H18N2O5 294.30
l-Phenylalanine, N-l- -aspartyl-, 1-methyl ester;
-aspartyl-, 1-methyl ester;
3-Amino-N-( -carboxyphenethyl)succinamic acid N-methyl ester
-carboxyphenethyl)succinamic acid N-methyl ester


 [22839-47-0].
[22839-47-0].
(as' par tame).
C14H18N2O5 294.30
l-Phenylalanine, N-l-
3-Amino-N-(
DEFINITION
Aspartame contains NLT 98.0% and NMT 102.0% of C14H18N2O5, calculated on the dried basis.
IDENTIFICATION
• A. Infrared Absorption  197M
197M
[Note—Do not dry specimens. ]
ASSAY
• Procedure
Sample:
300 mg
Titrimetric system
(See Titrimetry  541
541 .)
.)
0.1 N Perchloric acid:
Use perchloric acid, tenth-normal (0.1 N) in glacial acetic acid as specified for Reagents, Indicators, and Solutions—Volumetric Solutions, but in the standardization, titrate to a green endpoint.
Mode:
Direct titration
Titrant:
Use 0.1 N Perchloric acid
Blank:
1.5 mL of anhydrous formic acid and 60 mL of glacial acetic acid
Endpoint detection:
Visual
Analysis:
Dissolve the Sample in 1.5 mL of anhydrous formic acid, and add 60 mL of glacial acetic acid. Add crystal violet, and immediately titrate with the Titrant to a green endpoint. Perform a blank determination. [Note—A blank titration exceeding 0.1 mL may be due to excessive water content, and may cause loss of visual endpoint sensitivity. ]
Calculate the percentage of Aspartame (C14H18N2O5) in the Sample taken:
Result = [(V  B) × N × F × 100]/W
B) × N × F × 100]/W
| V | = | = volume of the Titrant consumed by the Sample (mL) |
| B | = | = volume of the Titrant consumed by the Blank (mL) |
| N | = | = actual normality of the Titrant (mEq/mL) |
| F | = | = equivalency factor, 294.3 mg/mEq |
| W | = | = weight of the Sample (mg) |
Acceptance criteria:
98.0%–102.0% on the dried basis
IMPURITIES
• Residue on Ignition  281
281 :
NMT 0.2%
:
NMT 0.2%
• Heavy Metals, Method II  231
231 :
NMT 10 ppm
:
NMT 10 ppm
• Limit of 5-Benzyl-3,6-dioxo-2-piperazineacetic Acid
Diluent:
Methanol and water (1:9)
Mobile phase:
Dissolve 5.6 g of monobasic potassium phosphate in 820 mL of water in a 1-L volumetric flask, adjust with phosphoric acid to a pH of 4.3, and dilute with methanol to volume.
Standard solution:
75 µg/mL of USP Aspartame Related Compound A RS in Diluent
Sample solution:
5 mg/mL of Aspartame in Diluent
[Note—Avoid heat and excessive holding times. ]
Chromatographic system
Mode:
LC
Detector:
UV 210 nm
Column:
4.6-mm × 25-cm; packing L1
Column temperature:
40
Flow rate:
2 mL/min
Injection size:
20 µL
System suitability
Sample:
Standard solution
Suitability requirements
Tailing factor:
NMT 2.0
Relative standard deviation:
NMT 4.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of aspartame related compound A in the portion of Aspartame taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of aspartame related compound A from the Sample solution |
| rS | = | = peak response of aspartame related compound A from the Standard solution |
| CS | = | = concentration of 5-benzyl-3,6-dioxo-2-piperazineacetic acid in the Standard solution (mg/mL) |
| CU | = | = concentration of Aspartame in the Sample solution (mg/mL) |
Acceptance criteria:
NMT 1.5%
• Chromatographic Purity
Diluent, Mobile phase, and Chromatographic system:
Proceed as directed in Limit of 5-Benzyl-3,6-dioxo-2-piperazineacetic Acid.
Sample stock solution:
Proceed as directed for Sample solution under Limit of 5-Benzyl-3,6-dioxo-2-piperazineacetic Acid.
Sample solution:
0.1 mg/mL of Aspartame from the Sample stock solution in Diluent
Analysis
Samples:
Sample stock solution and Sample solution
[Note—Continue the elution of Sample stock solution for twice the retention time of the aspartame peak. ]
Acceptance criteria:
The sum of the responses of all the peaks in the chromatogram of the Sample stock solution, excluding the 5-benzyl-3,6-dioxo-2-piperazineacetic acid and aspartame peak responses, is not greater than the aspartame peak response of the Sample solution, corresponding to NMT 2.0% of chromatographic impurities.
SPECIFIC TESTS
• Transmittance
Sample solution:
10 mg/mL of Aspartame in 2 N hydrochloric acid, prepared by means of sonication
Analysis:
Determine the transmittance in a 1-cm cell at 430 nm with a suitable spectrophotometer.
Acceptance criteria:
Transmittance of NLT 0.95, corresponding to an absorbance of NMT about 0.022
• Optical Rotation, Specific Rotation  781S
781S
Sample solution:
40 mg/mL in 15 N formic acid
Acceptance criteria:
+14.5 to +16.5
to +16.5 , determined at 20
, determined at 20 within 30 min after preparation of the Sample solution
within 30 min after preparation of the Sample solution
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 for 4 h: it loses NMT 4.5% of its weight.
for 4 h: it loses NMT 4.5% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well-closed containers.
• USP Reference Standards  11
11
USP Aspartame Related Compound A RS
5-Benzyl-3,6-dioxo-2-piperazineacetic acid.
C13H14N2O4 262.27
5-Benzyl-3,6-dioxo-2-piperazineacetic acid.
C13H14N2O4 262.27
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1702