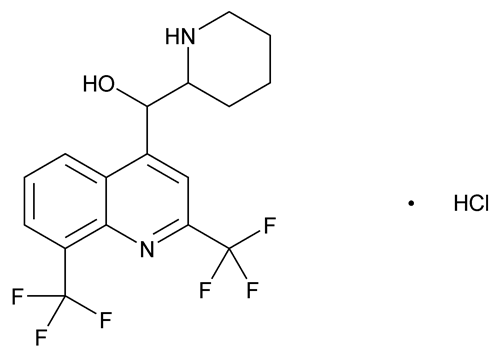
Mefloquine Hydrochloride
C17H16F6N2O·HCl 414.77
4-Quinolinemethanol, -2-piperidinyl-2,8-bis(trifluoromethyl)-, monohydrochloride, (R*,S*)- (±)-;
-2-piperidinyl-2,8-bis(trifluoromethyl)-, monohydrochloride, (R*,S*)- (±)-;
dl-erythro- -2-Piperidyl-2,8-bis(trifluoromethyl)-4-quinolinemethanol monohydrochloride
-2-Piperidyl-2,8-bis(trifluoromethyl)-4-quinolinemethanol monohydrochloride


 [51773-92-3].
[51773-92-3].
(me' floe kwin hye'' droe klor' ide).
C17H16F6N2O·HCl 414.77
4-Quinolinemethanol,
dl-erythro-
DEFINITION
Mefloquine Hydrochloride contains NLT 98.0% and NMT 102.0% of C17H16F6N2O·HCl, calculated on the anhydrous basis.
IDENTIFICATION
ASSAY
• Procedure
Solution A:
1.5 g/L of sodium hydrogen sulfate in water
Mobile phase:
Dissolve 1 g of tetraheptylammonium bromide in a 1000-mL mixture of acetonitrile, methanol, and Solution A (2:1:2).
System suitability solution:
4 µg/mL each of USP Mefloquine Hydrochloride RS and USP Mefloquine Related Compound A RS in Mobile phase
Standard solution:
0.2 mg/mL of USP Mefloquine Hydrochloride RS in Mobile phase
Sample solution:
0.2 mg/mL of Mefloquine Hydrochloride in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 280 nm
Guard column:
4-mm × 3-cm; C18 (recommended)
Column:
4.0-mm × 25-cm; 5-µm packing L1
Column temperature:
25
Flow rate:
0.8 mL/min
Injection size:
20 µL
System suitability
Samples:
System suitability solution and Standard solution
[Note—The relative retention times for mefloquine related compound A and mefloquine are about 0.7 and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 2.0 between mefloquine related compound A and mefloquine, System suitability solution
Tailing factor:
NMT 2.0, Standard solution
Relative standard deviation:
NMT 1.0%, Standard solution
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of mefloquine hydrochloride (C17H16F6N2O·HCl) in the portion of Mefloquine Hydrochloride taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of mefloquine from the Sample solution |
| rS | = | = peak response of mefloquine from the Standard solution |
| CS | = | = concentration of USP Mefloquine Hydrochloride RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Mefloquine Hydrochloride in the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0% on the anhydrous basis
IMPURITIES
• Residue on Ignition  281
281 :
NMT 0.1%
:
NMT 0.1%
• Heavy Metals, Method II  231
231 :
NMT 20 ppm
:
NMT 20 ppm
• Organic Impurities
Mobile phase:
Dissolve 1 g of tetraheptylammonium bromide in a 1-L mixture of a 1.5-g/L solution of sodium hydrogen sulfate, acetonitrile, and methanol (2:2:1).
System suitability solution:
4 µg/mL each of USP Mefloquine Hydrochloride RS and USP Mefloquine Related Compound A RS in Mobile phase. [Note—Mefloquine related compound A is threo-mefloquine. ]
Sample stock solution:
4 mg/mL of Mefloquine Hydrochloride in Mobile phase
Sample solution:
4 µg/mL from the Sample stock solution in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 280 nm
Guard column:
4-mm × 2.5-cm; 5-µm packing L1
Column:
4.0-mm × 25-cm; 5-µm packing L1
Flow rate:
0.8 mL/min
Injection size:
20 µL. [Note—Equilibrate the column with Mobile phase at a flow rate of 0.8 mL/min for 30 min. ]
System suitability
Sample:
System suitability solution
[Note—The relative retention times for mefloquine related compound A and mefloquine are about 0.7 and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 2.0 between mefloquine related compound A and mefloquine
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Sample stock solution and Sample solution
Record the chromatogram for a time that is 10 times the retention time of the main peak.
Acceptance criteria:
The response of the mefloquine related compound A peak in the Sample stock solution is NMT twice the area of the main peak of the Sample solution (0.2%). The response of any other individual peak, other than the main peak of the Sample stock solution, is NMT that of the main peak of the Sample solution (0.1%); and the sum of the responses of any such peaks of the Sample stock solution is NMT five times the response of the main peak of the Sample solution (0.5%). [Note—Exclude the main peak and any other peak producing a response of less than 0.2 times (0.02%) the main peak of the Sample solution. ]
SPECIFIC TESTS
• Optical Rotation, Specific Rotation  781S
781S
Sample solution:
50 mg/mL in methanol
Acceptance criteria:
–0.2 to +0.2
to +0.2
• Water Determination, Method I  921
921 :
NMT 3.0%
:
NMT 3.0%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight, light-resistant containers. Store between 15 and 30
and 30 .
.
• USP Reference Standards  11
11
USP Mefloquine Related Compound A RS
threo-Mefloquine.
C17H16F6N2O·HCl 414.78
threo-Mefloquine.
C17H16F6N2O·HCl 414.78
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Leonel M. Santos, Ph.D.
Senior Scientific Liaison 1-301-816-8168 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3783
Pharmacopeial Forum: Volume No. 35(5) Page 1160