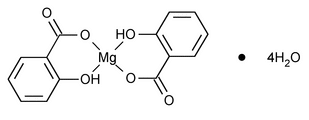
Magnesium Salicylate
(mag nee' zee um sa lis' i late).
Magnesium, bis(2-hydroxybenzoato-O1,O2)-, tetrahydrate.
Magnesium salicylate (1:2), tetrahydrate
Anhydrous 298.54
» Magnesium Salicylate contains not less than 98.0 percent and not more than 103.0 percent of C14H10MgO6·4H2O.
Packaging and storage—
Store in tight containers.
Identification—
A:
Infrared Absorption  197K
197K : previously dried at 105
: previously dried at 105 for 4 hours.
for 4 hours.
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
20 µg per mL.
Medium:
water.
C:
It responds to the test for Magnesium  191
191 .
.
Water, Method I  921
921 :
between 17.5% and 21.0%.
:
between 17.5% and 21.0%.
Heavy metals, Method I  231
231 :
0.004%.
:
0.004%.
Magnesium content—
Transfer about 800 mg, accurately weighed, to a 200-mL volumetric flask. Dissolve in and dilute with water to volume, and mix. Stir the resulting solution continuously for about 15 minutes and filter, discarding the first 10 mL of the filtrate, into a flask. Transfer 50.0 mL of the filtrate to a 250-mL conical flask, add 50 mL of water, 5 mL of ammonia–ammonium chloride buffer TS, and 0.15 mL of eriochrome black TS, and titrate with 0.05 M edetate disodium VS to a blue endpoint. Each mL of 0.05 M edetate disodium is equivalent to 1.215 mg of magnesium: between 6.3% and 6.7% of magnesium is found.
Assay—
Transfer 3.0 mL of the filtrate prepared in the test for Magnesium content to a 500-mL volumetric flask, dilute with water to volume, and mix. Concomitantly determine the absorbances of this solution and a solution of USP Salicylic Acid RS in water having a known concentration of about 18 µg per mL, in 1-cm cells at the wavelength of maximum absorbance at about 296 nm, with a suitable spectrophotometer, using water as the blank. Calculate the quantity, in mg, of C14H10MgO6·4H2O in the Magnesium Salicylate taken in the test for Magnesium content by the formula:
(370.59 / 276.24)(33.3C)(AU / AS)
in which 370.59 is the molecular weight of magnesium salicylate tetrahydrate; 276.24 is twice the molecular weight of salicylic acid; C is the concentration, in µg per mL, of USP Salicylic Acid RS in the Standard solution; and AU and AS are the absorbances of the test solution and the Standard solution, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Clydewyn M. Anthony, Ph.D.
Senior Scientific Liaison 1-301-816-8139 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3754