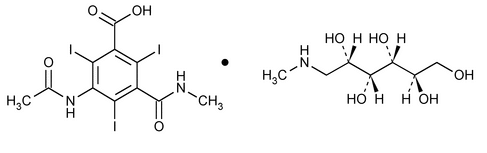Iothalamate Meglumine Injection
Benzoic acid, 3-(acetylamino)-2,4,6-triiodo-5-[(methylamino)carbonyl]-, compd. with 1-deoxy-1-(methylamino)-d-glucitol (1:1).
1-Deoxy-1-(methylamino)-d-glucitol 5-acetamido-2,4,6-triiodo-N-methylisophthalamate (salt)
» Iothalamate Meglumine Injection is a sterile solution of Iothalamic Acid in Water for Injection, prepared with the aid of Meglumine. It contains not less than 95.0 percent and not more than 105.0 percent of the labeled amount of iothalamate meglumine (C11H9I3N2O4·C7H17NO5). It may contain small amounts of suitable buffers and of Edetate Calcium Disodium or Edetate Disodium as a stabilizer. Iothalamate Meglumine Injection intended for intravascular use contains no antimicrobial agents.
Packaging and storage—
Preserve in single-dose containers, preferably of Type I glass, protected from light.
Labeling—
Label containers of Injection intended for intravascular injection to direct the user to discard any unused portion remaining in the container. Label containers of Injection intended for other than intravascular injection to show that the contents are not intended for intravascular injection.
USP Reference standards  11
11 —
—
USP 5-Amino-2,4,6-triiodo-N-methylisophthalamic Acid RS
C9H7I3N2O3 571.88
C9H7I3N2O3 571.88
USP Endotoxin RS
Identification—
Dilute 3 mL of Injection with water to 100 mL, add an excess of 3 N hydrochloric acid, and filter. Wash the precipitated iothalamic acid on the filter with four 10-mL portions of water, and dry at 105 for 4 hours: the dried iothalamic acid responds to the following tests.
for 4 hours: the dried iothalamic acid responds to the following tests.
A:
The IR absorption spectrum of a 0.5% potassium bromide dispersion of the dried acid exhibits maxima only at the same wavelengths as that of a similar preparation of USP Iothalamic Acid RS.
B:
Heat about 500 mg of the dried acid in a suitable crucible: violet vapors are evolved.
Bacterial endotoxins  85
85 —
It contains not more than 0.9 USP Endotoxin Unit per mL.
—
It contains not more than 0.9 USP Endotoxin Unit per mL.
pH  791
791 :
between 6.5 and 7.7.
:
between 6.5 and 7.7.
Free aromatic amine—
Dilute a suitable volume of Injection with water to yield a solution containing 100 mg of iothalamate meglumine per mL. Proceed as directed in the test for Free aromatic amine under Iothalamic Acid, beginning with “Pipet 5 mL of this solution into a 50-mL volumetric flask.”
Iodine and iodide—
Dilute a volume of Injection, equivalent to 2 g of iothalamate meglumine, with 20 mL of water in a 50-mL beaker, add 5 mL of 2 N sulfuric acid, stir, and filter into a glass-stoppered, 50-mL cylinder. Proceed as directed for Procedure in the test for Iodine and iodide under Iothalamic Acid, beginning with “To the filtrate add 5 mL of toluene.”
Heavy metals  231
231 —
In a 50-mL color-comparison tube, mix a volume of Injection, equivalent to 1.0 g of iothalamate meglumine, with 5 mL of 1 N sodium hydroxide, dilute with water to 40 mL, and mix. Using this as the Test preparation, proceed as directed in the test for Heavy metals under Diatrizoate Meglumine: the limit is 0.002%.
—
In a 50-mL color-comparison tube, mix a volume of Injection, equivalent to 1.0 g of iothalamate meglumine, with 5 mL of 1 N sodium hydroxide, dilute with water to 40 mL, and mix. Using this as the Test preparation, proceed as directed in the test for Heavy metals under Diatrizoate Meglumine: the limit is 0.002%.
Meglumine content—
Proceed as directed in the test for Meglumine content under Diatrizoate Meglumine Injection. The meglumine content is not less than 22.9% and not more than 25.3% of the labeled amount of iothalamate meglumine.
Other requirements—
It meets the requirements under Injections  1
1 .
.
Assay—
Pipet a volume of Injection, equivalent to about 4 g of iothalamate meglumine, into a 250-mL volumetric flask, dilute with water to volume, and mix. Pipet 25 mL of this solution into a glass-stoppered, 125-mL conical flask, add 12 mL of 5 N sodium hydroxide and 1 g of powdered zinc, connect the flask to a reflux condenser, and reflux for 30 minutes. Cool to room temperature, rinse the condenser with 20 mL of water, disconnect the flask from the condenser, and filter the mixture. Rinse the filter and the flask thoroughly, adding the rinsings to the filtrate. Add 40 mL of 2 N sulfuric acid, and titrate immediately with 0.05 N silver nitrate VS, determining the endpoint potentiometrically, using silver-calomel electrodes and an agar-potassium nitrate salt bridge. Each mL of 0.05 N silver nitrate is equivalent to 13.49 mg of C11H9I3N2O4·C7H17NO5.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
|
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology |
USP35–NF30 Page 3544