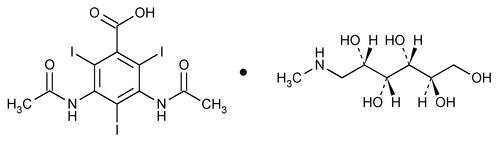Diatrizoate Meglumine
(dye'' a trye zoe' ate me' gloo meen).
Benzoic acid, 3,5-bis(acetylamino)-2,4,6-triiodo-, compd. with 1-deoxy-1-(methylamino)-d-glucitol (1:1).
1-Deoxy-1-(methylamino)-d-glucitol 3,5-diacetamido-2,4,6-triiodobenzoate (salt)
» Diatrizoate Meglumine contains not less than 98.0 percent and not more than 102.0 percent of C11H9I3N2O4·C7H17NO5, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers. Store at 25 , excursions permitted between 15
, excursions permitted between 15 and 30
and 30 .
.
Identification—
A:
It responds to the Thin-Layer Chromatographic Identification Test  201
201 , the test solution and the Standard solution of USP Diatrizoic Acid RS being prepared at a concentration of 1 mg per mL in a 0.8 in 1000 solution of sodium hydroxide in methanol, the solvent mixture being a mixture of chloroform, methanol, and ammonium hydroxide (20:10:2), and short-wavelength UV light being used to locate the spots.
, the test solution and the Standard solution of USP Diatrizoic Acid RS being prepared at a concentration of 1 mg per mL in a 0.8 in 1000 solution of sodium hydroxide in methanol, the solvent mixture being a mixture of chloroform, methanol, and ammonium hydroxide (20:10:2), and short-wavelength UV light being used to locate the spots.
B:
Heat about 500 mg in a suitable crucible: violet vapors are evolved.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 4 hours: it loses not more than 1.0% of its weight.
for 4 hours: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Iodine and iodide—
Test preparation—
Transfer 2.0 g to a 50-mL centrifuge tube provided with a stopper, dilute with water to 24 mL, and shake to dissolve.
Procedure—
Add 5 mL of toluene and 5 mL of 2 N sulfuric acid, shake, and centrifuge: the toluene layer shows no red color. Add 1 mL of sodium nitrite solution (1 in 50), shake, and centrifuge: any red color in the toluene layer is not darker than that obtained when a mixture of 2.0 mL of potassium iodide solution (1 in 4000) and 22 mL of water is substituted for the solution under test (0.02% of iodide).
Heavy metals  231
231 —
—
Standard preparation—
Transfer 2.0 mL of Standard Lead Solution (20 µg of Pb) to a 50-mL color-comparison tube, add 5 mL of 1 N sodium hydroxide, dilute with water to 40 mL, and mix.
Test preparation—
Dissolve 1.0 g of Diatrizoate Meglumine in 20 mL of water and 5 mL of 1 N sodium hydroxide, transfer the solution to a 50-mL color-comparison tube, dilute with water to 40 mL, and mix.
Procedure—
To each of the tubes containing the Standard preparation and the Test preparation add 10 mL of sodium sulfide TS, mix, allow to stand for 5 minutes, and view downward over a white surface: the color of the solution from the Test preparation is not darker than that of the solution from the Standard preparation (0.002%).
Free aromatic amine—
Transfer 1.0 g to a 50-mL volumetric flask, and add 5 mL of water and 10 mL of 0.1 N sodium hydroxide. To a second 50-mL volumetric flask transfer 4 mL of water, 10 mL of 0.1 N sodium hydroxide, and 1.0 mL of a Standard solution prepared by dissolving a suitable quantity of USP Diatrizoic Acid Related Compound A RS in 0.1 N sodium hydroxide. Use 0.2 mL of 0.1 N sodium hydroxide for each 5.0 mg of Standard, and dilute with water to obtain a known concentration of 500 µg per mL. To a third 50-mL volumetric flask add 5 mL of water and 10 mL of 0.1 N sodium hydroxide to provide a blank.
Treat each flask as follows. Add 25 mL of dimethyl sulfoxide, insert the stopper, and mix by swirling gently. Chill in an ice bath in the dark for 5 minutes. [note—In conducting the following steps, keep the flasks in the ice bath and in the dark as much as possible until all of the reagents have been added. ] Slowly add 2 mL of hydrochloric acid, mix, and allow to stand for 5 minutes. Add 2 mL of sodium nitrite solution (1 in 50), mix, and allow to stand for 5 minutes. Add 1 mL of sulfamic acid solution (2 in 25), shake, and allow to stand for 5 minutes. [Caution—Considerable pressure is produced.
] Add 2 mL of a 1 in 1000 solution of N-(1-naphthyl)-ethylenediamine dihydrochloride in dilute propylene glycol (7 in 10), and mix.
Remove the flasks from the ice bath and from storage in the dark, and allow to stand in a water bath at 22 to 25
to 25 for 10 minutes. Shake gently and occasionally during this period, releasing the pressure by loosening the stopper. Dilute with water to volume, and mix.
for 10 minutes. Shake gently and occasionally during this period, releasing the pressure by loosening the stopper. Dilute with water to volume, and mix.
Within 5 minutes from the time of diluting the solutions in all three flasks to 50 mL, concomitantly determine the absorbances of the solution from the substance under test and the Standard solution in 1-cm cells at the wavelength of maximum absorbance at about 465 nm, with a suitable spectrophotometer, versus the prepared blank. The absorbance of the solution from the Diatrizoate Meglumine is not greater than that of the Standard solution (0.05%).
Assay—
Transfer about 400 mg of Diatrizoate Meglumine, accurately weighed, to a glass-stoppered, 125-mL conical flask, add 30 mL of 1.25 N sodium hydroxide and 500 mg of powdered zinc, connect the flask to a reflux condenser, and reflux the mixture for 1 hour. Cool the flask to room temperature, rinse the condenser with 20 mL of water, disconnect the flask from the condenser, and filter the mixture. Rinse the flask and the filter thoroughly, adding the rinsings to the filtrate. Add 5 mL of glacial acetic acid and 1 mL of tetrabromophenolphthalein ethyl ester TS, and titrate with 0.05 N silver nitrate VS until the yellow precipitate just turns green. Each mL of 0.05 N silver nitrate is equivalent to 13.49 mg of C11H9I3N2O4·C7H17NO5.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2863
Pharmacopeial Forum: Volume No. 30(3) Page 832