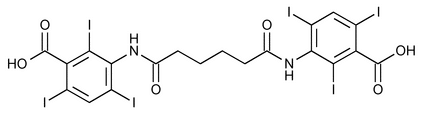
Iodipamide
(eye'' oh dip' a mide).
Benzoic acid, 3,3¢-[(1,6-dioxo-1,6-hexanediyl)diimino]bis[2,4,6-triiodo-.
3,3¢-(Adipoyldiimino)bis[2,4,6-triiodobenzoic acid]
» Iodipamide contains not less than 98.0 percent and not more than 102.0 percent of C20H14I6N2O6, calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed containers. Store at 25 , excursions permitted between 15
, excursions permitted between 15 and 30
and 30 .
.
Identification—
A:
It responds to the Thin-Layer Chromatographic Identification Test  201
201 , the test solution and the Standard solution being prepared at a concentration of 1 mg per mL in an 0.8 in 1000 solution of sodium hydroxide in methanol, the solvent mixture being a mixture of chloroform, methanol, and ammonium hydroxide (20:10:2), and short-wavelength UV light being used to locate the spots.
, the test solution and the Standard solution being prepared at a concentration of 1 mg per mL in an 0.8 in 1000 solution of sodium hydroxide in methanol, the solvent mixture being a mixture of chloroform, methanol, and ammonium hydroxide (20:10:2), and short-wavelength UV light being used to locate the spots.
B:
Heat about 500 mg in a suitable crucible: violet vapors are evolved.
Water, Method I  921
921 :
not more than 1.0%.
:
not more than 1.0%.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Free aromatic amine—
Transfer 1.0 g to a 50-mL volumetric flask, and add 12.5 mL of water and 2.5 mL of 1 N sodium hydroxide. To a second 50-mL volumetric flask transfer 4 mL of water, 10 mL of 0.1 N sodium hydroxide, and 1.0 mL of a Standard solution prepared by dissolving a suitable quantity of USP 3-Amino-2,4,6-triiodobenzoic Acid RS in 0.1 N sodium hydroxide (use 0.2 mL of the 0.1 N sodium hydroxide for each 5.0 mg of the Reference standard) and by diluting with water to obtain a solution having a known concentration of 500 µg per mL. Proceed as directed in the test for Free aromatic amine under Diatrizoate Meglumine, beginning with “To a third 50-mL volumetric flask add 5 mL of water.”
Other requirements—
It meets the requirements of the tests for Iodine and iodide and Heavy metals under Diatrizoic Acid.
Assay—
Transfer about 300 mg of Iodipamide, accurately weighed, to a glass-stoppered, 125-mL conical flask, add 30 mL of 1.25 N sodium hydroxide and 500 mg of powdered zinc, connect the flask to a reflux condenser, and reflux the mixture for 30 minutes. Cool the flask to room temperature, rinse the condenser with 20 mL of water, disconnect the flask from the condenser, and filter the mixture. Rinse the filter and the flask thoroughly, adding the rinsings to the filtrate. Add 5 mL of glacial acetic acid and 1 mL of tetrabromophenolphthalein ethyl ester TS, and titrate with 0.05 N silver nitrate VS until the yellow precipitate just turns green. Each mL of 0.05 N silver nitrate is equivalent to 9.498 mg of C20H14I6N2O6.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3525
Pharmacopeial Forum: Volume No. 29(6) Page 1907