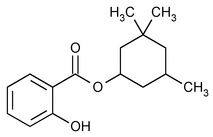
Homosalate
(hoe'' moe sal' ate).
Benzoic acid, 2-hydroxy, 3,3,5-trimethylcyclohexyl ester.
3,3,5-Trimethylcyclohexyl salicylate
» Homosalate contains not less than 90.0 percent and not more than 110 percent of C16H22O3, calculated on the as-is basis.
Packaging and storage—
Preserve in tight containers.
Identification, Infrared Absorption  197F
197F .
.
Specific gravity  841
841 :
between 1.049 and 1.053.
:
between 1.049 and 1.053.
Refractive index  831
831 :
between 1.516 and 1.519 at 20
:
between 1.516 and 1.519 at 20 .
.
Assay—
Standard preparation—
Dissolve an accurately weighed quantity of USP Homosalate RS in chloroform, and dilute quantitatively, and stepwise if necessary, with chloroform to obtain a solution having a known concentration of about 5.0 mg per mL.
Assay preparation—
Dissolve about 50 mg of Homosalate, accurately weighed, in 10 mL of chloroform, and mix.
Chromatographic system (see Chromatography  621
621 )—
The gas chromatograph is equipped with a flame-ionization detector and a 0.53-mm × 30-m column coated with a 1-µm film of G27. Hydrogen is used as the carrier gas at a flow rate of about 6 mL per minute. The chromatograph is programmed as follows. Initially the temperature of the column is equilibrated at 70
)—
The gas chromatograph is equipped with a flame-ionization detector and a 0.53-mm × 30-m column coated with a 1-µm film of G27. Hydrogen is used as the carrier gas at a flow rate of about 6 mL per minute. The chromatograph is programmed as follows. Initially the temperature of the column is equilibrated at 70 ; upon injection the temperature is increased at a rate of 6
; upon injection the temperature is increased at a rate of 6 per minute to 220
per minute to 220 , and is held at 220
, and is held at 220 for 5 minutes. The injection port temperature is maintained at 270
for 5 minutes. The injection port temperature is maintained at 270 , and the detector is maintained at 270
, and the detector is maintained at 270 . Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the relative retention times are about 0.98 for the cis-isomer and 1.0 for the trans-isomer; the resolution, R, between the cis-isomer and the trans-isomer is not less than 1.0; and the relative standard deviation for replicate injections is not more than 2.0%.
. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the relative retention times are about 0.98 for the cis-isomer and 1.0 for the trans-isomer; the resolution, R, between the cis-isomer and the trans-isomer is not less than 1.0; and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 1 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of C16H22O3 in the portion of Homosalate taken by the formula:
10C(rU / rS)
in which C is the concentration, in mg per mL, of USP Homosalate RS in the Standard preparation; and rU and rS are the sums of the peak responses for the cis- and trans-isomers obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Feiwen Mao, M.S.
Senior Scientific Liaison 1-301-816-8320 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3417
Pharmacopeial Forum: Volume No. 37(2)