Glyceryl Distearate
(glis' er il dye steer' ate).
625.01


 [1323-83-7].
[1323-83-7].
» Glyceryl Distearate is a mixture of diglycerides, mainly glyceryl distearate, together with variable quantities of monoglycerides and triglycerides. It contains not less than 8.0 percent and not more than 22.0 percent of monoglycerides, not less than 40.0 percent and not more than 60.0 percent of diglycerides, and not less than 25.0 percent and not more than 35.0 percent of triglycerides. It is obtained by partial glycerolysis of vegetable oil that consists mainly of triglycerides of palmitic or stearic acid or by esterification of glycerol with stearic acid. The fatty acids may be of vegetable or animal origin.
Packaging and storage—
Preserve in tight containers. No storage requirements specified.
Identification—
A:
It meets the requirements of the test for Melting range.
B:
Thin-Layer Chromatographic Identification Test  201
201 —
—
Test solution:
a solution in methylene chloride containing about 50 mg per mL.
Developing solvent:
a mixture of ether and hexane (70:30).
Spray reagent:
a solution containing 20 mg of rhodamine B in 200 mL of alcohol.
Procedure—
Proceed as directed in the chapter. Spray with the Spray reagent, and locate the spots on the plate by examination under UV light at a wavelength of 365 nm: the principal spot obtained from the Test solution corresponds in color, size, and RF value to that obtained from the Standard solution.
Melting range, Class II  741
741 :
between 50
:
between 50 and 70
and 70 .
.
Acid value  401
401 :
not more than 6.0, determined on 1.0 g, using a mixture of alcohol and toluene (1:1, v/v) as the solvent and with gentle heating.
:
not more than 6.0, determined on 1.0 g, using a mixture of alcohol and toluene (1:1, v/v) as the solvent and with gentle heating.
Iodine value  401
401 :
not more than 3.0.
:
not more than 3.0.
Saponification value  401
401 :
between 165 and 195, determined on 2.0 g; carry out the titration with heating.
:
between 165 and 195, determined on 2.0 g; carry out the titration with heating.
Fatty acid composition  401
401 —
The fatty acid fraction of it contains not less than 40.0% of stearic acid; and the sum of the contents of palmitic and stearic acids, determined as directed in the chapter, is not less than 90.0%.
—
The fatty acid fraction of it contains not less than 40.0% of stearic acid; and the sum of the contents of palmitic and stearic acids, determined as directed in the chapter, is not less than 90.0%.
Water, Method I  921
921 :
not more than 1.0%, using pyridine in place of methanol in the titration vessel.
:
not more than 1.0%, using pyridine in place of methanol in the titration vessel.
Total ash  561
561 :
not more than 0.1%.
:
not more than 0.1%.
Limit of nickel—
[Caution:When using closed high-pressure digestion vessels and microwave laboratory equipment, be familiar with the safety and operating instructions given by the manufacturer.
]
Magnesium nitrate solution—
Transfer 2.5 g of magnesium nitrate to a 500-mL volumetric flask, and dissolve in and dilute with water to volume.
Test solution—
Transfer an accurately weighed quantity of about 0.1 g of Glyceryl Distearate into a suitable high-pressure resistant digestion vessel (fluoropolymer or quartz), and add 6.0 mL of nitric acid and 2.0 mL of 30% hydrogen peroxide. Place the closed vessel in a laboratory microwave oven, and digest using an appropriate program (e.g., 250 W for 10 minutes; 600 W for 5 minutes; 400 W for 5 minutes; and 250 W for 7 minutes.) Allow the digestion vessel to cool before opening. Quantitatively transfer the contents to a 25-mL volumetric flask, dilute with water to volume, and mix.
Blank solution—
Add 6.0 mL of nitric acid and 2.0 mL of 30% hydrogen peroxide to a high-pressure-resistant digestion vessel and proceed as directed for the Test solution.
Calibration solutions—
Transfer 5.0 mL of nickel standard solution TS to a 10-mL volumetric flask, add 0.5 mL of nitric acid and 1.0 mL of 30 percent hydrogen peroxide, and dilute with water to volume (stock solution). Into four identical 25-mL volumetric flasks, each containing 6 mL of nitric acid, transfer respectively 20, 50, 100, and 150 µL of the stock solution obtained above, and dilute with water to volume. These solutions contain 4 ng per mL, 10 ng per mL, 20 ng per mL, and 30 ng per mL of nickel, respectively.
Zero solution—
Transfer 6.0 mL of nitric acid to a 25-mL volumetric flask, add a small amount of water, mix, and then dilute with water to volume.
Procedure—
Into seven separate 25-mL flasks, each containing 5.0 mL of Magnesium nitrate solution, transfer respectively 10.0 mL of each of the following: the Test solution, the Blank solution, the four Calibration solutions, and the Zero solution. Concomitantly determine the absorbances of these solutions at least three times each, at the wavelength of maximum absorbance at 232.0 nm, with a suitable atomic absorption spectrophotometer (see Spectrophotometry and Light-Scattering  851
851 ) equipped with a pyrolytically coated tube with a platform, a graphite furnace, and a nickel hollow-cathode lamp. Maintain the drying temperature of the furnace at 100
) equipped with a pyrolytically coated tube with a platform, a graphite furnace, and a nickel hollow-cathode lamp. Maintain the drying temperature of the furnace at 100 for 10 seconds after a 10-second ramp, the ashing temperature at 1400
for 10 seconds after a 10-second ramp, the ashing temperature at 1400 for 10 seconds after a 20-second ramp, and the atomization temperature at 2500
for 10 seconds after a 20-second ramp, and the atomization temperature at 2500 for 5 seconds. Use the Zero solution to set the instrument to zero. Record the average of the steady readings for each of the solutions [note—If necessary, dilute the Test solution with the Zero solution to obtain a reading within the calibrated absorbance range ] : not more than 1 µg per g is found.
for 5 seconds. Use the Zero solution to set the instrument to zero. Record the average of the steady readings for each of the solutions [note—If necessary, dilute the Test solution with the Zero solution to obtain a reading within the calibrated absorbance range ] : not more than 1 µg per g is found.
Limit of free glycerin—
Mobile phase and Chromatographic system—
Proceed as directed in the Assay.
Standard solutions—
Prepare four solutions by dissolving accurately weighed quantities of glycerin in tetrahydrofuran, and diluting each with tetrahydrofuran, as necessary, to obtain solutions having known concentrations of about 0.2, 0.4, 1.0, and 2.0 mg per mL.
Test solution—
Use the Assay preparation, prepared as directed in the Assay.
Procedure—
Separately inject equal volumes (about 40 µL) of the Standard solutions and the Test solution into the chromatograph, record the chromatograms, and measure the responses for the glycerin peaks. Plot the glycerin peak responses obtained versus the concentration, in mg per mL, of glycerin in the Standard solutions. From the standard curve so obtained, determine the glycerin concentration, C, in mg per mL, in the Test solution. Calculate the percentage of free glycerin in the portion of Glyceryl Distearate taken by the formula:
500(C/W)
in which C is as obtained above; and W is the amount, in mg, of Glyceryl Distearate taken to prepare the Test solution: not more than 1.0% of free glycerin is found.
Assay—
Mobile phase:
tetrahydrofuran.
Assay preparation—
Transfer about 200 mg of Glyceryl Distearate, accurately weighed, to a 5-mL volumetric flask, dissolve in and dilute with tetrahydrofuran to volume, and mix.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a refractive index detector and a 7.5-mm × 60-cm column containing 5-µm 100-
)—
The liquid chromatograph is equipped with a refractive index detector and a 7.5-mm × 60-cm column containing 5-µm 100-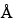 packing L21. The column and the detector temperatures are maintained at 40
packing L21. The column and the detector temperatures are maintained at 40 . [Note—Two or three 7.5-mm × 30-cm L21 columns may be used in place of the one 60-cm column, provided that system suitability requirements are met; and the column temperature may be lowered to ambient temperature, although working at 40
. [Note—Two or three 7.5-mm × 30-cm L21 columns may be used in place of the one 60-cm column, provided that system suitability requirements are met; and the column temperature may be lowered to ambient temperature, although working at 40 provides stable separation conditions and ensures better sample solubility. ] The flow rate is about 1 mL per minute. Chromatograph the Assay preparation, and record the peak responses as directed for Procedure: the relative retention times are about 1.0 for glycerin, 0.84 for monoglycerides, 0.78 for diglycerides, and 0.75 for triglycerides; the resolution, R, between the diglycerides and monoglycerides is not less than 1.0; and the relative standard deviation for replicate injections determined from the monoglycerides peak is not more than 2.0%.
provides stable separation conditions and ensures better sample solubility. ] The flow rate is about 1 mL per minute. Chromatograph the Assay preparation, and record the peak responses as directed for Procedure: the relative retention times are about 1.0 for glycerin, 0.84 for monoglycerides, 0.78 for diglycerides, and 0.75 for triglycerides; the resolution, R, between the diglycerides and monoglycerides is not less than 1.0; and the relative standard deviation for replicate injections determined from the monoglycerides peak is not more than 2.0%.
Procedure—
Inject a volume (about 40 µL) of the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the percentage of monoglycerides, diglycerides, and triglycerides in the portion of Glyceryl Distearate taken by the formula:
100(ri / rs)
in which ri is the individual peak response for the monoglycerides, diglycerides, and triglycerides, as appropriate; and rs is the sum of the responses for all of the glyceride peaks.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1812
Pharmacopeial Forum: Volume No. 30(3) Page 975
