Powdered Garlic
DEFINITION
Powdered Garlic is produced from Garlic that has been cut, freeze-dried or dried at a temperature not exceeding 65 , and powdered. It contains NLT 0.3% of alliin and NLT 0.1% of
, and powdered. It contains NLT 0.3% of alliin and NLT 0.1% of 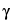 -glutamyl-(S)-allyl-l-cysteine, calculated on the dried basis.
-glutamyl-(S)-allyl-l-cysteine, calculated on the dried basis.
IDENTIFICATION
• A. Thin-Layer Chromatographic Identification Test
Standard solution A:
0.5 mg/mL of USP l-Methionine RS in a mixture of methanol and water (1:1)
Standard solution B:
0.5 mg/mL of USP Alliin RS in a mixture of methanol and water (1:1)
Sample solution:
Transfer 1 g of the Powdered Garlic to an extractor, and extract with two 20-mL portions of a mixture of methanol and water (1:1), combining the extracts. Concentrate to a small volume (about 5 mL), using a rotary evaporator.
Adsorbent:
0.25-mm layer of chromatographic silica gel, typically 20 cm long (TLC plates)
Application volume:
20 µL, applied separately as 10-mm bands
Developing solvent system:
Butyl alcohol, n-propyl alcohol, glacial acetic acid, and water (3:1:1:1)
Spray reagent:
0.2 in 100 solution of ninhydrin in a mixture of butyl alcohol and 2 N acetic acid (19:1)
Analysis
Samples:
Standard solution A, Standard solution B, and Sample solution
Develop the chromatograms until the solvent front has moved up about three-fourths of the plate, in a saturated chamber. Remove the plate, and allow the solvent to evaporate. Spray with Spray reagent, heat at 100 –105
–105 for 10 min, and immediately examine the plate.
for 10 min, and immediately examine the plate.
Acceptance criteria:
The chromatogram of the Sample solution shows the following orange and pinkish violet zones: a violet zone having an RF value of about 0.89; a pink zone having an RF value of about 0.5 and corresponding in color and RF value to that obtained from the chromatogram of Standard solution A; a pinkish zone having an RF value of about 0.43; a strong orange zone having an RF value of about 0.38; a pinkish violet zone having an RF value of about 0.3 and corresponding in color and RF value to that of the chromatogram of Standard solution B; and additional pinkish orange zones situated very close to each other just below the zone attributed to alliin in the chromatogram of Standard solution B.
• B.
Sample:
About 10 g of Powdered Garlic
Analysis:
Transfer the Sample to a suitable flask. Add 10 mL of 1 N sodium hydroxide and 10 mL of water, heat the flask in boiling water for 10 min, cool, and filter. Add a few drops of freshly prepared sodium nitroferricyanide TS to 2 mL of the filtrate.
Acceptance criteria:
The appearance of a red or orange-red color indicates the presence of sulfur-containing compounds in the Sample.
• C.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the test for Content of Alliin.
• D. Thin-Layer Chromatographic Identification Test
Extraction column:
1-cm × 5-cm solid-phase extraction column; contains styrene-divinylbenzene copolymer packing with a 75- to 150-µm diameter and a 400- to 600- pore size. Condition column before use by washing with 50 mL of methanol and with 50 mL of a mixture of methanol and water (3:7). [Note—Do not allow the column to dry. ]
pore size. Condition column before use by washing with 50 mL of methanol and with 50 mL of a mixture of methanol and water (3:7). [Note—Do not allow the column to dry. ]
Standard solution:
0.2 mg/mL each of USP  -Chlorogenin RS and USP Agigenin RS in methanol
-Chlorogenin RS and USP Agigenin RS in methanol
Sample solution:
Transfer about 10 g of the Powdered Garlic to a 37-mL homogenizing cup, and homogenize with 25 mL of methanol at the highest speed for 1 min. Centrifuge the mixture, and decant the supernatant to a flask. Add 70 mL of water. Transfer to the Extraction column, allow to drain, and discard the eluate. Wash the column with 50 mL of a mixture of methanol and water (3:7), allow the solvent mixture to drain, and discard the eluate. Finally, elute the crude saponin fraction on the column with 20 mL of methanol, and collect the eluate. Evaporate the solvent to dryness. Dissolve the residue in 4 mL of a mixture of 8% sulfuric acid and alcohol (1:1), transfer the solution to a screw-capped test tube, and heat on a boiling water bath for 5 h. Cool the test tube, add 20 mL of water, and transfer the solution to a freshly conditioned Extraction column, allow to drain, and discard the eluate. Wash the column with 30 mL of a mixture of methanol and water (7:3), and discard the eluate. Finally, elute the column with 50 mL of methanol. Collect the eluate, evaporate it to dryness, and dissolve the residue in 0.5 mL of methanol.
Adsorbent:
0.25-mm layer of chromatographic silica gel, typically 20 cm long (TLC plates)
Application volume:
20 µL, as 7-mm bands
Developing solvent system:
Methylene chloride and methanol (15:2)
Spray reagent:
Dissolve 0.5 mL of 4-methoxybenzaldehyde and 0.5 mL of sulfuric acid in sufficient alcohol to make 10 mL.
Analysis
Samples:
Standard solution and Sample solution
Develop the chromatograms until the solvent front has moved up about three-fourths of the plate, in a saturated chamber. Remove the plate, and allow the solvent to evaporate. Spray the plate with Spray reagent, heat the plate at 100 –105
–105 for 5 min, and examine the plate.
for 5 min, and examine the plate.
Acceptance criteria:
The chromatogram of the Sample solution exhibits, among several yellowish and grayish green spots, a grayish green spot at an RF value of about 0.4, corresponding to the grayish green spot due to  -chlorogenin of the Standard solution. The chromatogram of the Sample solution exhibits no spot at an RF value of about 0.2, corresponding to agigenin of the Standard solution.
-chlorogenin of the Standard solution. The chromatogram of the Sample solution exhibits no spot at an RF value of about 0.2, corresponding to agigenin of the Standard solution.
COMPOSITION
• Content of Alliin
Alliinase inhibitor solution:
Dissolve 109 mg of carboxymethoxylamine hemihydrochloride in 100.0 mL of water.
Solution A:
0.045 M monobasic sodium phosphate in water. Adjust with 0.2 M sodium hydroxide to a pH of 7.1.
Buffer:
0.05 M monobasic sodium phosphate in water. Adjust with 0.2 M sodium hydroxide to a pH of 9.5.
Derivatization reagent:
Dissolve 140 mg of o-phthaldialdehyde in 5 mL of methanol, add 100 µL of t-butylthiol, and dilute with Buffer to 50 mL. [Note—This reagent may occasionally become opaque during preparation. Store at room temperature, and use within 1 week. ]
Mobile phase:
Acetonitrile, 1,4-dioxane, tetrahydrofuran, and Solution A (25: 2.9: 2.2: 69.9)
Standard solution:
0.05 mg/mL of USP Alliin RS in a mixture of methanol and water (1:1)
Sample stock solution:
Transfer about 1.0 g of Powdered Garlic, accurately weighed, to a flask. Add 30.0 mL of Alliinase inhibitor solution, and shake vigorously until the powder is fully dispersed. Centrifuge to obtain a clear solution.
Sample solution:
Transfer 5.0 mL of the Sample stock solution to a 10-mL volumetric flask, and dilute with Alliinase inhibitor solution to volume.
Chromatographic system
Mode:
LC
Detector:
UV 337 nm
Column:
4-mm × 10-cm; packing L1
Flow rate:
1 mL/min
Injection size:
10 µL
System suitability
Sample:
Standard solution
Suitability requirements
[Note—Alliin exhibits two major peaks representing its diastereomers. ]
Relative standard deviation:
NMT 2.0% for each of the major peaks, in repeated injections
Analysis
Samples:
Standard solution and Sample solution
Using a syringe, transfer 0.1 mL of the Standard solution or the Sample solution to separate septum-capped vials, add 0.5 mL of the Derivatization reagent to each vial, and mix. Allow a reaction time of NLT 2 min before injection into the chromatograph. Record the chromatograms, and measure the areas of the alliin diastereomer peaks.
Calculate the percentage of alliin in the portion of Powdered Garlic taken:
Result = (rU/rS) × CS × (V/W) × D × 100
| rU | = | = peak area of alliin from the Sample solution |
| rS | = | = sum of the peak areas of alliin diastereomers from the Standard solution |
| CS | = | = concentration of USP Alliin RS in the Standard solution (mg/mL) |
| V | = | = volume of the Sample stock solution (mL) |
| W | = | = weight of Powdered Garlic used to prepare the Sample stock solution (mg) |
| D | = | = dilution factor to prepare the Sample solution from the Sample stock solution, 2 |
Acceptance criteria:
NLT 0.3% on the dried basis
• Content of 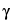 -Glutamyl-(S)-allyl-l-cysteine
-Glutamyl-(S)-allyl-l-cysteine
Solution A:
Dissolve 6.80 g of monobasic potassium phosphate in 900 mL of water, and adjust with phosphoric acid to a pH of 2.6. Dilute with water to 1000.0 mL, and mix.
Mobile phase:
Methanol and Solution A (3:17)
Standard solution:
0.08 mg/mL of USP 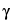 -Glutamyl-(S)-allyl-l-cysteine RS in a mixture of methanol and water (1:1)
-Glutamyl-(S)-allyl-l-cysteine RS in a mixture of methanol and water (1:1)
Sample solution:
Transfer about 1.0 g of Powdered Garlic, accurately weighed, to a 50-mL volumetric flask. Add 30 mL of methanol and water (1:1), and shake vigorously until the powder is fully dispersed. Dilute the contents of the flask with a mixture of methanol and water (1:1) to volume. Centrifuge to obtain a clear solution.
Chromatographic system
Mode:
LC
Detector:
UV 205 nm
Column:
4.6-mm × 15-cm; packing L1
Flow rate:
0.8 mL/min
Injection size:
10 µL
System suitability
Sample:
Standard solution
Suitability requirements
Relative standard deviation:
NMT 2.0% for the 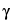 -glutamyl-(S)-allyl-l-cysteine peak in repeated injections
-glutamyl-(S)-allyl-l-cysteine peak in repeated injections
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of 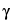 -glutamyl-(S)-allyl-l-cysteine in the portion of Powdered Garlic taken:
-glutamyl-(S)-allyl-l-cysteine in the portion of Powdered Garlic taken:
Result = (rU/rS) × CS × (V/W) × 100
| rU | = | = peak response for |
| rS | = | = peak response for |
| CS | = | = concentration of USP |
| V | = | = volume of the Sample solution (mL) |
| W | = | = weight of Powdered Garlic used to prepare the Sample solution (mg) |
Acceptance criteria:
NLT 0.1% on the dried basis
CONTAMINANTS
• Heavy Metals, Method I  231
231 :
NMT 10 ppm
:
NMT 10 ppm
• Articles of Botanical Origin, Pesticide Residues  561
561 :
Meets the requirements
:
Meets the requirements
SPECIFIC TESTS
• Botanic Characteristics:
Under a microscope, Powdered Garlic shows the following diagnostic characteristics: numerous fragments of parenchyma with large cells containing crystals of calcium oxalate and small triangular or quadrangular intercellular spaces at the corners; spiral vessels accompanied by subquadratic cells; elongated epidermal cells with thick, pitted walls.
• Absence of Starch:
A water slurry of Powdered Garlic shows no blue color when iodine TS is added.
• Loss on Drying  731
731 :
Dry 1 g of it at 105
:
Dry 1 g of it at 105 for 2 h: it loses NMT 7.0% of its weight.
for 2 h: it loses NMT 7.0% of its weight.
• Articles of Botanical Origin, Total Ash  561
561 :
NMT 5.0%
:
NMT 5.0%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Store in well-closed containers in a cool, dry place, protected from light.
• Labeling:
The label states the Latin binomial and, following the official name, the part of the plant source from which the article was derived.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Maged H. Sharaf, Ph.D.
Principal Scientific Liaison 1-301-816-8318 |
(DS2010) Monographs - Dietary Supplements |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1311
