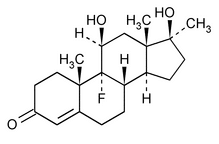
Fluoxymesterone
(floo ox'' i mes' ter one).
Androst-4-en-3-one, 9-fluoro-11,17-dihydroxy-17-methyl-, (11
9-Fluoro-11
» Fluoxymesterone contains not less than 97.0 percent and not more than 102.0 percent of C20H29FO3, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers, protected from light.
Identification—
A:
Infrared Absorption  197K
197K —If a difference appears, dissolve portions of both the test specimen and the USP Reference Standard in dehydrated alcohol, evaporate the solutions to dryness, and repeat the test on the residues.
—If a difference appears, dissolve portions of both the test specimen and the USP Reference Standard in dehydrated alcohol, evaporate the solutions to dryness, and repeat the test on the residues.
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
10 µg per mL.
Medium:
alcohol.
Absorptivities at 242 nm do not differ by more than 2.5%.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 3 hours: it loses not more than 1.0% of its weight.
for 3 hours: it loses not more than 1.0% of its weight.
Chromatographic purity—
Solution A—
Prepare a filtered and degassed mixture of methanol and water (55:45).
Solution B—
Use filtered and degassed methanol.
Mobile phase—
Use variable mixtures of Solution A and Solution B as directed for Chromatographic system.
Blank solution—
Use Solution B.
System suitability solution—
Dilute a volume of the Test solution quantitatively, and stepwise if necessary, with methanol to obtain a solution having a concentration of about 5 µg of fluoxymesterone per mL.
Test solution—
Prepare a solution of Fluoxymesterone in Solution B containing about 0.5 mg per mL.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 254-nm detector and a 4.6-mm × 25-cm column that contains packing L1. The column temperature is maintained at 40
)—
The liquid chromatograph is equipped with a 254-nm detector and a 4.6-mm × 25-cm column that contains packing L1. The column temperature is maintained at 40 . The flow rate is 1.0 mL per minute. The chromatograph is programmed as follows.
. The flow rate is 1.0 mL per minute. The chromatograph is programmed as follows.
Chromatograph the Test solution, and record the peak responses as directed for Procedure: the column efficiency is not less than 15,000 theoretical plates. Chromatograph the System suitability solution, and record the peak responses as directed for Procedure: the signal-to-noise ratio for the fluoxymesterone peak is not less than 100.
| Time (minutes) |
Solution A
(%) |
Solution B
(%) |
Elution |
|---|---|---|---|
| 0 | 100 | 0 | equilibration |
| 0–20 | 100®60 | 0®40 | linear gradient |
| 20–40 | 60®0 | 40®100 | linear gradient |
| 40–45.0 | 0 | 100 | isocratic |
| 45.0–45.1 | 0®100 | 100®0 | linear gradient |
| 45.1–60 | 100 | 0 | isocratic |
Procedure—
Separately inject equal volumes (about 5 µL) of the Blank solution and the Test solution into the chromatograph, record the chromatograms, and measure the areas for any peaks that do not appear in the Blank solution that have an area equal to or greater than 0.1% of the fluoxymesterone peak. Calculate the percentage of each impurity in the portion of Fluoxymesterone taken by the formula:
100(ri / rs)
in which ri is the peak response for each impurity; and rs is the sum of the responses of all the peaks: not more than 1.0% of any individual impurity is found; and not more than 2.0% of total impurities is found.
Assay—
Internal standard solution—
Dissolve methylprednisolone in a mixture of chloroform and methanol (95:5) to obtain a solution containing about 200 µg per mL.
Mobile phase—
Prepare a solution containing butyl chloride, water-saturated butyl chloride, tetrahydrofuran, methanol, and glacial acetic acid (475:475:70:35:30).
Standard preparation—
Dissolve an accurately weighed quantity of USP Fluoxymesterone RS in Internal standard solution to obtain a solution having a known concentration of about 0.25 mg per mL.
Assay preparation—
Dissolve about 25 mg of Fluoxymesterone, accurately weighed, in 100.0 mL of Internal standard solution to obtain a solution having a concentration of about 0.25 mg per mL.
Procedure—
Inject equal volumes of the Assay preparation and the Standard preparation into a suitable high-pressure liquid chromatograph (see Chromatography  621
621 ) of the general type equipped with a detector for monitoring UV light at 254 nm, equipped with a suitable recorder, and capable of providing column pressure up to about 2000 psi. The instrument contains a 4-mm × 30-cm stainless steel column that contains packing L3. In a suitable chromatogram, the resolution, R, between fluoxymesterone and the internal standard is not less than 3.0; and the relative standard deviation of the peak response ratios of four replicate injections of the Standard preparation is not more than 2.0%. Calculate the quantity, in mg, of C20H29FO3 in the portion of Fluoxymesterone taken by the formula:
) of the general type equipped with a detector for monitoring UV light at 254 nm, equipped with a suitable recorder, and capable of providing column pressure up to about 2000 psi. The instrument contains a 4-mm × 30-cm stainless steel column that contains packing L3. In a suitable chromatogram, the resolution, R, between fluoxymesterone and the internal standard is not less than 3.0; and the relative standard deviation of the peak response ratios of four replicate injections of the Standard preparation is not more than 2.0%. Calculate the quantity, in mg, of C20H29FO3 in the portion of Fluoxymesterone taken by the formula:
100C(RU / RS)
in which C is the concentration, in mg per mL, of USP Fluoxymesterone RS in the Standard preparation; and RU and RS are the peak response ratios of fluoxymesterone to the internal standard obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Domenick Vicchio, Ph.D.
Senior Scientific Liaison 1-301-998-6828 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3247
Pharmacopeial Forum: Volume No. 28(1) Page 59