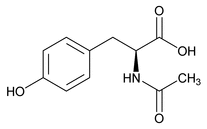
N-Acetyltyrosine
C11H13NO4 223.2
N-Acetyl-l-tyrosine;
(2S)-2-(Acetylamino)-3-(4-hydroxyphenyl)propanoic acid)

 [537-55-3].
[537-55-3].
C11H13NO4 223.2
N-Acetyl-l-tyrosine;
(2S)-2-(Acetylamino)-3-(4-hydroxyphenyl)propanoic acid)
DEFINITION
N-Acetyltyrosine contains NLT 98.5% and NMT 101.0% of C11H13NO4, as N-acetyl-l-tyrosine, calculated on the dried basis.
IDENTIFICATION
• B. Optical Rotation, Specific Rotation  781S
781S
Sample solution:
10 mg/mL
Acceptance criteria:
NLT +46.0 and NMT +49.0
and NMT +49.0 , determined at 20
, determined at 20
• C.
The RF value of the principal spot of the Sample solution in the test for Organic Impurities corresponds to that of Standard solution 1.
ASSAY
• Procedure
Sample solution:
Dissolve about 180 mg of N-Acetyltyrosine, weighed, in 50 mL of carbon dioxide-free water.
Titrimetric system
(See Titrimetry  541
541 .)
.)
Mode:
Direct titration
Titrant:
0.1 N sodium hydroxide VS
Endpoint detection:
Potentiometric
Equivalency:
Each mL of 0.1 N sodium hydroxide VS is equivalent to 22.32 mg of C11H13NO4.
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.1%
:
NMT 0.1%
• Chloride and Sulfate, Chloride  221
221 :
NMT 200 ppm. A 0.7-g portion shows no more chloride than corresponds to 0.40 mL of 0.01 N hydrochloric acid.
:
NMT 200 ppm. A 0.7-g portion shows no more chloride than corresponds to 0.40 mL of 0.01 N hydrochloric acid.
• Chloride and Sulfate, Sulfate  221
221 :
NMT 200 ppm. A 1.2-g portion shows no more sulfate than corresponds to 0.25 mL of 0.020 N sulfuric acid.
:
NMT 200 ppm. A 1.2-g portion shows no more sulfate than corresponds to 0.25 mL of 0.020 N sulfuric acid.
• Iron  241
241 :
NMT 20 ppm
:
NMT 20 ppm
• Heavy Metals, Method 1  231
231 :
NMT 10 ppm
:
NMT 10 ppm
Organic Impurities
• Procedure
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture
Standard stock solution 1:
8 mg/mL of USP N-Acetyl-l-tyrosine RS in a mixture of water, glacial acetic acid, and alcohol (3:3:94)
Standard solution 1:
Dilute Standard stock solution 1 with alcohol to obtain a solution having a known concentration of about 0.4 mg/mL.
Standard solution 2:
0.8 mg/mL of USP l-Tyrosine RS dissolved in a mixture of glacial acetic acid and water (1:1), and diluted with alcohol
Sample solution:
Transfer 0.8 g of N-Acetyltyrosine to a 10-mL volumetric flask, dissolve in 6 mL of a mixture of glacial acetic acid and water (1:1), and dilute with alcohol to volume.
Application volume:
5 µL
Developing solvent system:
A mixture of ammonia and 2-propanol (3:7)
Spray reagent:
Dissolve 0.2 g of ninhydrin in 100 mL of a mixture of butanol and 2 N acetic acid (95:5).
Analysis:
Proceed as directed for Chromatography  621
621 , Thin-Layer Chromatography. After air-drying the plate, repeat the development process. After air-drying a second time, spray with Spray reagent, and heat between 100
, Thin-Layer Chromatography. After air-drying the plate, repeat the development process. After air-drying a second time, spray with Spray reagent, and heat between 100 and 105
and 105 for about 15 min.
for about 15 min.
Acceptance criteria:
Any secondary spot from the Sample solution is not larger or more intense than the principal spot from Standard solution 1, except for the spot corresponding to tyrosine, which is not larger or more intense than the principal spot from Standard solution 2.
Individual impurities:
NMT 0.5%
Limit of tyrosine:
NMT 1.0%
SPECIFIC TESTS
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 for 3 h: it loses NMT 0.1% of its weight.
for 3 h: it loses NMT 0.1% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well-closed containers, and store at controlled room temperature.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Huy T. Dinh, M.S.
Scientific Liaison 1-301-816-8594 |
(DS2010) Monographs - Dietary Supplements |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1174
Pharmacopeial Forum: Volume No. 35(1) Page 107