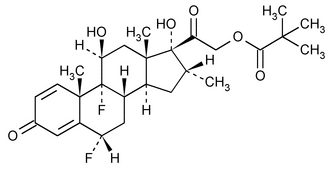
Flumethasone Pivalate
(floo meth' a sone piv' a late).
Pregna-1,4-diene-3,20-dione, 21-(2,2-dimethyl-1-oxopropoxy)-6,9-difluoro-11,17-dihydroxy-16-methyl-, (6
6
» Flumethasone Pivalate contains not less than 97.0 percent and not more than 103.0 percent of C27H36F2O6, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
20 µg per mL.
Medium:
methanol.
Absorptivities at 237 nm, calculated on the dried basis, do not differ by more than 3.0%.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 4 hours: it loses not more than 1.0% of its weight.
for 4 hours: it loses not more than 1.0% of its weight.
Chromatographic purity—
Prepare a solution in dioxane containing 20 mg per mL. Apply 5 µL of this solution and 5 µL each of three dioxane solutions containing in each mL, respectively, 200 (1%), 400 (2%), and 600 (3%) µg of USP Flumethasone Pivalate RS to a suitable thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel. Allow the spots to dry, and develop the chromatogram in a solvent system consisting of toluene and ethyl acetate (7:3) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow the solvent to evaporate. Locate the spots on the plate by lightly spraying with dilute sulfuric acid (1 in 2), heating at 100
) coated with a 0.25-mm layer of chromatographic silica gel. Allow the spots to dry, and develop the chromatogram in a solvent system consisting of toluene and ethyl acetate (7:3) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow the solvent to evaporate. Locate the spots on the plate by lightly spraying with dilute sulfuric acid (1 in 2), heating at 100 for 30 minutes, and inspecting under long-wavelength UV light: the total content of any impurities detected, when compared to the Standard solutions, does not exceed 3.0%.
for 30 minutes, and inspecting under long-wavelength UV light: the total content of any impurities detected, when compared to the Standard solutions, does not exceed 3.0%.
Assay—
Standard preparation—
Dissolve a suitable quantity of USP Flumethasone Pivalate RS, accurately weighed, in alcohol, and dilute quantitatively and stepwise with alcohol to obtain a solution having a known concentration of about 20 µg per mL. Transfer 10.0 mL of this solution to a glass-stoppered, 20-mL conical flask.
Assay preparation—
Transfer about 20 mg of Flumethasone Pivalate, accurately weighed, to a 100-mL volumetric flask, dissolve in and dilute with alcohol to volume, and mix. Transfer 10.0 mL of this solution to a second 100-mL volumetric flask, dilute with alcohol to volume, and mix. Transfer 10.0 mL of this solution to a glass-stoppered, 20-mL conical flask.
Procedure—
To each of the flasks containing the Standard preparation and the Assay preparation, and to a similar flask containing 10.0 mL of alcohol to provide the blank, add 1.0 mL of tetramethylammonium hydroxide TS. Mix, allow to stand for 20 minutes, accurately timed, add 1.0 mL of blue tetrazolium TS to each flask, and mix. Allow to stand for 40 minutes, add 1.0 mL of glacial acetic acid to each flask, mix, and concomitantly determine the absorbances of the solutions in 1-cm cells at the wavelength of maximum absorbance at about 520 nm, with a suitable spectrophotometer, against the blank. Calculate the quantity, in mg, of C27H36F2O6 in the portion of Flumethasone Pivalate taken by the formula:
C(AU / AS)
in which C is the concentration, in µg per mL, of USP Flumethasone Pivalate RS in the Standard preparation; and AU and AS are the absorbances of the solutions from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Domenick Vicchio, Ph.D.
Senior Scientific Liaison 1-301-998-6828 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3220