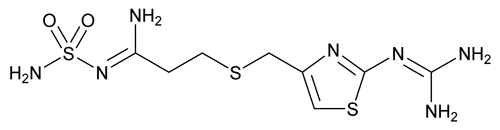
Famotidine
(fam oh' ti deen).
Propanimidamide, N¢-(aminosulfonyl)-3-[[[2-[(diaminomethylene)amino]-4-thiazolyl]methyl]thio]-.
[1-Amino-3-[[[2-[(diaminomethylene)amino]-4-thiazolyl]methyl]thio]propylidene] sulfamide
» Famotidine contains not less than 98.5 percent and not more than 101.0 percent of C8H15N7O2S3, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers, protected from light.
Identification—
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
25 µg per mL.
Medium:
phosphate buffer.
Absorptivities at 265 nm, calculated on the dried basis, do not differ by more than 3.0%. [note—Prepare the phosphate buffer as follows. Adjust 250 mL of 0.02 M phosphoric acid with sodium hydroxide solution (1 in 10) to a pH of 2.5, dilute with water to 500 mL, and mix. ]
Loss on drying  731
731 —
Dry it at a pressure between 1 and 5 mm of mercury at 80
—
Dry it at a pressure between 1 and 5 mm of mercury at 80 for 5 hours: it loses not more than 0.5% of its weight.
for 5 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method II  231
231 :
not more than 0.001%.
:
not more than 0.001%.
Chromatographic purity—
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture.
Test solution—
Transfer about 200 mg of Famotidine, accurately weighed, to a 10-mL volumetric flask, add 2 mL of methanol, and shake for 10 minutes. Add 0.1 mL of glacial acetic acid, stir until dissolved, dilute with methanol to volume, and mix.
Standard solutions—
Dissolve an accurately weighed portion of USP Famotidine RS in a mixture of methanol and glacial acetic acid (100:1) to obtain Standard solution 1 having a known concentration of 0.2 mg per mL. Dilute a portion of this solution, accurately measured, with a mixture of methanol and glacial acetic acid (100:1) to obtain Standard solution 2 containing 65 µg of USP Famotidine RS per mL.
Developing solvent system:
a mixture of ethyl acetate, methanol, toluene, and ammonium hydroxide (40:25:20:2).
Procedure—
Separately apply 5 µL of the Test solution and 5 µL of each Standard solution to a plate, and dry under a stream of nitrogen. Proceed as directed for Thin-Layer Chromatography under Chromatography  621
621 . Examine the plate under short-wavelength UV light, and compare the intensities of any secondary spots observed in the chromatogram of the Test solution with those of the principal spots in the chromatograms of the Standard solutions: no secondary spot from the chromatogram of the Test solution is larger in size or more intense than the principal spot obtained from Standard solution 2 (0.3%); and the sum of the intensities of the secondary spots obtained from the Test solution corresponds to not more than 1.0% (Standard solution 1).
. Examine the plate under short-wavelength UV light, and compare the intensities of any secondary spots observed in the chromatogram of the Test solution with those of the principal spots in the chromatograms of the Standard solutions: no secondary spot from the chromatogram of the Test solution is larger in size or more intense than the principal spot obtained from Standard solution 2 (0.3%); and the sum of the intensities of the secondary spots obtained from the Test solution corresponds to not more than 1.0% (Standard solution 1).
Assay—
Dissolve about 250 mg of Famotidine, accurately weighed, in 80 mL of glacial acetic acid, and titrate with 0.1 N perchloric acid VS (see Titrimetry  541
541 ), using a suitable anhydrous electrode system. Any aqueous electrolyte solution contained in the electrodes employed should be removed, the electrode rendered anhydrous and filled with 0.1 N lithium perchlorate in acetic anhydride. Perform a blank determination and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 16.87 mg of C8H15N7O2S3.
), using a suitable anhydrous electrode system. Any aqueous electrolyte solution contained in the electrodes employed should be removed, the electrode rendered anhydrous and filled with 0.1 N lithium perchlorate in acetic anhydride. Perform a blank determination and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 16.87 mg of C8H15N7O2S3.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Elena Gonikberg, Ph.D.
Principal Scientific Liaison 1-301-816-8251 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3150
Pharmacopeial Forum: Volume No. 37(3)