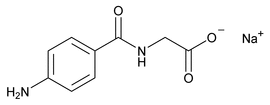
Aminohippurate Sodium Injection
Glycine, N-(4-aminobenzoyl)-, monosodium salt.
Monosodium p-aminohippurate
» Aminohippurate Sodium Injection is a sterile solution of Aminohippuric Acid in Water for Injection prepared with the aid of Sodium Hydroxide. It contains not less than 95.0 percent and not more than 105.0 percent of the labeled amount of C9H9N2NaO3.
Packaging and storage—
Preserve in single-dose or multiple-dose containers, preferably of Type I glass.
USP Reference standards  11
11 —
—
USP Endotoxin RS
Identification—
A:
A volume of Injection, equivalent to 100 mg of aminohippuric acid, diluted to 50 mL and acidified with hydrochloric acid, responds to Identification test B under Aminohippuric Acid.
B:
Transfer a volume of Injection, equivalent to about 200 mg of aminohippurate sodium, to a test tube, and add, in the order named, 2 mL of potassium iodide TS, 10 mL of water, and 5 mL of sodium hypochlorite TS: a red color is produced.
C:
It responds to the flame test for Sodium  191
191 .
.
Bacterial endotoxins  85
85 —
It contains not more than 0.04 USP Endotoxin Unit per mg of aminohippurate sodium.
—
It contains not more than 0.04 USP Endotoxin Unit per mg of aminohippurate sodium.
pH  791
791 :
between 6.7 and 7.6.
:
between 6.7 and 7.6.
Other requirements—
It meets the requirements under Injections  1
1 .
.
Assay—
Transfer to a 200-mL volumetric flask an accurately measured volume of Injection, equivalent to about 1 g of aminohippurate sodium, and dilute with water to volume. Transfer 50.0 mL of the solution to a suitable container, add 5 mL of hydrochloric acid, and proceed as directed under Nitrite Titration  451
451 , beginning with “cool to about 15
, beginning with “cool to about 15 .” Each mL of 0.1 M sodium nitrite is equivalent to 21.62 mg of C9H9N2NaO3.
.” Each mL of 0.1 M sodium nitrite is equivalent to 21.62 mg of C9H9N2NaO3.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Thomas A. Sigambris, M.S.
Scientific Liaison 1-301-998-6789 |
(BIO12010) Monographs - Biologics and Biotechnology 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
|
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology |
USP35–NF30 Page 2171