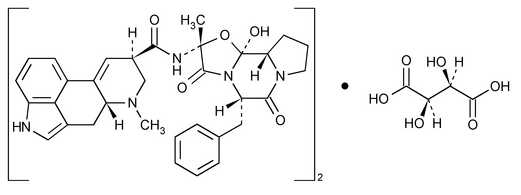
Ergotamine Tartrate
(er got' a meen tar' trate).
Ergotaman-3¢,6¢,18-trione, 12¢-hydroxy-2¢-methyl-5¢-(phenylmethyl)-, (5¢
Ergotamine tartrate (2:1) (salt)
» Ergotamine Tartrate contains not less than 97.0 percent and not more than 100.5 percent of (C33H35 N5O5)2·C4H6O6, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers in a cold place.
Identification—
The chromatogram of the test solution prepared as directed in the test for Related alkaloids exhibits its principal fluorescent spot and principal blue spot at the same RF value as the principal spot of Standard solution A.
Specific rotation  781S
781S —
[note—For this test, use chloroform from which any alcohol present has been removed by prior washing with water. ] Dissolve about 350 mg in 25 mL of tartaric acid solution (1 in 100) contained in a separator, then add 500 mg of sodium bicarbonate, and mix gently but thoroughly. Add 10 mL of chloroform, shake vigorously, and after the layers have separated draw off the chloroform phase through a small filter, previously moistened with chloroform, into a 50-mL volumetric flask. Rapidly continue the extraction with three 10-mL portions of chloroform, passing the extracts through the same filter. Place the flask in a bath at 20
—
[note—For this test, use chloroform from which any alcohol present has been removed by prior washing with water. ] Dissolve about 350 mg in 25 mL of tartaric acid solution (1 in 100) contained in a separator, then add 500 mg of sodium bicarbonate, and mix gently but thoroughly. Add 10 mL of chloroform, shake vigorously, and after the layers have separated draw off the chloroform phase through a small filter, previously moistened with chloroform, into a 50-mL volumetric flask. Rapidly continue the extraction with three 10-mL portions of chloroform, passing the extracts through the same filter. Place the flask in a bath at 20 for 10 minutes. Adjust the volume of extract to 50.0 mL at 20
for 10 minutes. Adjust the volume of extract to 50.0 mL at 20 by the addition of chloroform. Mix the solution, and determine the angular rotation at 20
by the addition of chloroform. Mix the solution, and determine the angular rotation at 20 . Determine the concentration of ergotamine in the chloroform solution by evaporating a 25.0-mL aliquot of the solution on a rotary evaporator to dryness, maintaining the temperature of the bath below 45
. Determine the concentration of ergotamine in the chloroform solution by evaporating a 25.0-mL aliquot of the solution on a rotary evaporator to dryness, maintaining the temperature of the bath below 45 . Dissolve the residue in 25 mL of glacial acetic acid, add 1 drop of crystal violet TS, and titrate with 0.05 N perchloric acid VS to an emerald-green endpoint. Perform a blank determination, and make any necessary correction. Each mL of 0.05 N perchloric acid is equivalent to 29.08 mg of C33H35N5O5. From the angular rotation of the solution and the concentration of ergotamine base, calculate the specific rotation of the base: between
. Dissolve the residue in 25 mL of glacial acetic acid, add 1 drop of crystal violet TS, and titrate with 0.05 N perchloric acid VS to an emerald-green endpoint. Perform a blank determination, and make any necessary correction. Each mL of 0.05 N perchloric acid is equivalent to 29.08 mg of C33H35N5O5. From the angular rotation of the solution and the concentration of ergotamine base, calculate the specific rotation of the base: between  155
155 and
and  165
165 .
.
Loss on drying  731
731 —
Dry about 100 mg, accurately weighed, in vacuum at 60
—
Dry about 100 mg, accurately weighed, in vacuum at 60 for 4 hours: it loses not more than 5.0% of its weight.
for 4 hours: it loses not more than 5.0% of its weight.
Related alkaloids—
[note—Conduct this test without exposure to daylight and with the minimum necessary exposure to artificial light. ]
Solvent mixture—
Mix 9 volumes of chloroform with 1 volume of methanol.
Standard preparation
and Standard dilutions—Prepare a solution of USP Ergotamine Tartrate RS in Solvent mixture to contain 10.0 mg per mL (Standard preparation). Prepare a series of dilutions of the Standard preparation in Solvent mixture to contain 0.2 mg, 0.1 mg, 0.05 mg, and 0.025 mg per mL (Standard dilutions) corresponding to 2.0%, 1.0%, 0.5%, and 0.25% of the Standard preparation, respectively.
Test preparation—
Dissolve 50.0 mg of Ergotamine Tartrate in 5.0 mL of Solvent mixture.
Procedure—
In a suitable chromatographic chamber arranged for thin-layer chromatography (see Chromatography  621
621 ) place a volume of a solvent system consisting of ether, dimethylformamide, chloroform, and dehydrated alcohol (70:15:10:5). Line the chamber with filter paper, and allow it to equilibrate for 15 minutes. Apply 5-µL portions of the Test preparation, the Standard preparation, and each of the Standard dilutions to a suitable chromatographic plate coated with a 0.25-mm layer of chromatographic silica gel. Place each spot over an opened bottle of ammonium hydroxide for 20 seconds, then allow the plate to dry in a current of cold air for 20 seconds. Develop the chromatogram until the solvent front has moved about 17 cm. Remove the plate from the developing chamber, allow the solvent to evaporate in a current of cold air for approximately 2 minutes, and spray with a freshly prepared solution of 200 mg of p-(dimethylamino)benzaldehyde in a mixture of 5.5 mL of hydrochloric acid and 4.5 mL of water. Dry the plate at 60
) place a volume of a solvent system consisting of ether, dimethylformamide, chloroform, and dehydrated alcohol (70:15:10:5). Line the chamber with filter paper, and allow it to equilibrate for 15 minutes. Apply 5-µL portions of the Test preparation, the Standard preparation, and each of the Standard dilutions to a suitable chromatographic plate coated with a 0.25-mm layer of chromatographic silica gel. Place each spot over an opened bottle of ammonium hydroxide for 20 seconds, then allow the plate to dry in a current of cold air for 20 seconds. Develop the chromatogram until the solvent front has moved about 17 cm. Remove the plate from the developing chamber, allow the solvent to evaporate in a current of cold air for approximately 2 minutes, and spray with a freshly prepared solution of 200 mg of p-(dimethylamino)benzaldehyde in a mixture of 5.5 mL of hydrochloric acid and 4.5 mL of water. Dry the plate at 60 for about 5 minutes and compare the chromatograms: the RF value of the principal spot obtained from the Test preparation corresponds to that obtained from the Standard preparation, the sum of the intensities of any secondary spots in the chromatogram from the Test preparation is not greater than the intensity of the principal spot from the 2.0% Standard dilution, and the intensity of not more than one of the secondary spots is greater than that of the principal spot from the 1.0% Standard dilution.
for about 5 minutes and compare the chromatograms: the RF value of the principal spot obtained from the Test preparation corresponds to that obtained from the Standard preparation, the sum of the intensities of any secondary spots in the chromatogram from the Test preparation is not greater than the intensity of the principal spot from the 2.0% Standard dilution, and the intensity of not more than one of the secondary spots is greater than that of the principal spot from the 1.0% Standard dilution.
Assay—
Transfer about 200 mg of Ergotamine Tartrate, accurately weighed, to a small conical flask, and dissolve in 15 mL of a mixture of 6 volumes of acetic anhydride and 100 volumes of glacial acetic acid. Add 1 drop of crystal violet TS, and titrate with 0.05 N perchloric acid VS from a 10-mL buret. Perform a blank determination, and make any necessary correction. Each mL of 0.05 N perchloric acid is equivalent to 32.84 mg of (C33H35N5O5)2·C4H6O6.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3072
Pharmacopeial Forum: Volume No. 29(6) Page 1884