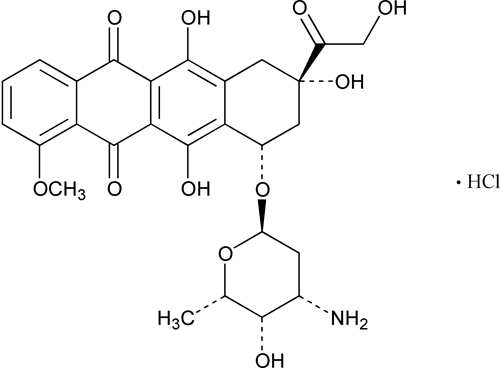
Doxorubicin Hydrochloride
(dox'' oh roo' bi sin hye'' droe klor' ide).
5,12-Naphthacenedione, 10-[(3-amino-2,3,6-trideoxy-
(8S,10S)-10-[3-Amino-2,3,6-trideoxy-
» Doxorubicin Hydrochloride contains not less than 98.0 percent and not more than 102.0 percent of C27H29NO11·HCl, calculated on the anhydrous, solvent-free basis.
[Caution—Great care should be taken to prevent inhaling particles of doxorubicin hydrochloride and exposing the skin to it.
]
Packaging and storage—
Preserve in tight containers, and store at controlled room temperature, except where it is labeled as amorphous, in which case it should be stored in the freezer.
Labeling—
The amorphous form is so labeled.
Identification—
The retention time of the doxorubicin peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
Crystallinity  695
695 :
meets the requirements, except that where it is labeled as amorphous, most of the particles do not exhibit birefringence and extinction positions.
:
meets the requirements, except that where it is labeled as amorphous, most of the particles do not exhibit birefringence and extinction positions.
pH  791
791 :
between 4.0 and 5.5, in a solution containing 5 mg per mL.
:
between 4.0 and 5.5, in a solution containing 5 mg per mL.
Water, Method I  921
921 :
not more than 4.0%.
:
not more than 4.0%.
Chromatographic purity—
Proceed as directed in the Assay, except to use, instead of the Assay preparation, a test solution prepared by dissolving Doxorubicin Hydrochloride in Mobile phase to obtain a solution containing about 0.5 mg per mL. From the chromatogram of the test solution, calculate the percentage of impurities taken by the formula:
100S/(S + r)
in which S is the sum of the responses of the minor component peaks; and r is the response of the major doxorubicin peak: the total of any impurities detected is not more than 2.0%.
Limit of solvent residues (as acetone and alcohol)—
Standard solution—
Transfer to a 100-mL volumetric flask about 200 mg of acetone, 300 mg of dehydrated alcohol, and 1000 mg of dioxane, each accurately weighed, and mix. Dilute with water to volume, and mix. Transfer 5.0 mL of the resulting solution to a 50-mL volumetric flask, dilute with water to volume, and mix. This solution contains about 0.2 mg of acetone, 0.3 mg of C2H5OH, and 1 mg of dioxane per mL.
Solvent—
Transfer about 100 mg of dioxane, accurately weighed, to a 100-mL volumetric flask, dilute with water to volume, and mix.
Test solution—
Dissolve about 200 mg of Doxorubicin Hydrochloride in 3.0 mL (3.0 g) of Solvent.
Chromatographic system (see Chromatography  621
621 )—
The gas chromatograph is equipped with a flame-ionization detector and a 4-mm × 2-m column packed with 8% to 10% liquid phase G16 and 2% potassium hydroxide on 100- to 120-mesh support S1A. The column temperature is maintained at about 60
)—
The gas chromatograph is equipped with a flame-ionization detector and a 4-mm × 2-m column packed with 8% to 10% liquid phase G16 and 2% potassium hydroxide on 100- to 120-mesh support S1A. The column temperature is maintained at about 60 , and helium is used as the carrier gas. Adjust the column temperature and carrier gas flow rate so that dioxane elutes in about 6 minutes. Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the relative retention times are about 0.2 for acetone, 0.5 for alcohol, and 1.0 for dioxane; the resolution, R, between adjacent peaks is not less than 2.0; the tailing factor for the alcohol peak is not more than 1.5; and the relative standard deviations of the ratios of the peak responses of the acetone and dioxane peaks and of the alcohol and dioxane peaks for replicate injections is not more than 4.0%.
, and helium is used as the carrier gas. Adjust the column temperature and carrier gas flow rate so that dioxane elutes in about 6 minutes. Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the relative retention times are about 0.2 for acetone, 0.5 for alcohol, and 1.0 for dioxane; the resolution, R, between adjacent peaks is not less than 2.0; the tailing factor for the alcohol peak is not more than 1.5; and the relative standard deviations of the ratios of the peak responses of the acetone and dioxane peaks and of the alcohol and dioxane peaks for replicate injections is not more than 4.0%.
Procedure—
Separately inject equal volumes (about 1 µL) of the Standard solution and the Test solution into the chromatograph, record the chromatograms, and measure the areas for the major peaks. Calculate the percentage, by weight, of acetone (CH3COCH3) and alcohol (C2H5OH), respectively, in the portion of Doxorubicin Hydrochloride taken by the same formula:
100(CA / CD)(DU / WU)(RU / RS)
in which CA is the concentration, in mg per mL, of acetone or alcohol in the Standard solution; CD is the concentration, in mg per mL, of dioxane in the Standard solution; DU is the total quantity, in mg, of dioxane in the Test solution; WU is the quantity, in mg, of Doxorubicin Hydrochloride taken to prepare the Test solution; and RU and RS are the area ratios of the analyte peak (acetone or alcohol) to the dioxane peak obtained from the Test solution and the Standard solution, respectively: not more than 0.5% of acetone is found; and the total of acetone and alcohol is not greater than 2.5%. Use the combined percentage of acetone and alcohol obtained to calculate the result obtained as directed in the Assay on the solvent-free basis.
Assay—
Mobile phase—
Prepare a suitable mixture of water, acetonitrile, methanol, and phosphoric acid (540:290:170:2). Dissolve 1 g of sodium lauryl sulfate in 1000 mL of this solution, adjust with 2 N sodium hydroxide to a pH of 3.6 ± 0.1, and degas. Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Resolution solution—
Dissolve about 10 mg of Doxorubicin Hydrochloride in 5 mL of water, add 5 mL of phosphoric acid, and allow to stand for about 30 minutes. Adjust with 2 N sodium hydroxide (about 37 mL) to a pH of 2.6 ± 0.1, add 15 mL of acetonitrile and 10 mL of methanol, mix, and filter. [note—Portions of this solution may be frozen until needed, then thawed, and mixed before use. ]
Standard preparation—
Dissolve an accurately weighed quantity of USP Doxorubicin Hydrochloride RS in Mobile phase to obtain a solution having a known concentration of about 0.1 mg per mL.
Assay preparation—
Transfer about 20 mg of Doxorubicin Hydrochloride, accurately weighed, to a 200-mL volumetric flask, add Mobile phase to volume, and mix.
Chromatographic system (see Chromatography  621
621 )—
The chromatograph is equipped with a 254-nm detector and a 4.6-mm × 25-cm column that contains packing L13. The flow rate is about 1.5 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the tailing factor for the doxorubicin peak is not less than 0.7 and not more than 1.2; the column efficiency, determined from the doxorubicin peak, is not less than 2250 theoretical plates; and the relative standard deviation for replicate injections is not more than 1.0%. Chromatograph the Resolution solution, and record the peak responses as directed for Procedure: the relative retention times are about 0.6 for doxorubicinone and 1.0 for doxorubicin; and the resolution, R, between doxorubicinone and doxorubicin is not less than 5.5.
)—
The chromatograph is equipped with a 254-nm detector and a 4.6-mm × 25-cm column that contains packing L13. The flow rate is about 1.5 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the tailing factor for the doxorubicin peak is not less than 0.7 and not more than 1.2; the column efficiency, determined from the doxorubicin peak, is not less than 2250 theoretical plates; and the relative standard deviation for replicate injections is not more than 1.0%. Chromatograph the Resolution solution, and record the peak responses as directed for Procedure: the relative retention times are about 0.6 for doxorubicinone and 1.0 for doxorubicin; and the resolution, R, between doxorubicinone and doxorubicin is not less than 5.5.
Procedure—
Separately inject equal volumes (about 20 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of C27H29NO11·HCl in the Doxorubicin Hydrochloride taken by the formula:
0.2CP(rU / rS)
in which C is the concentration, in mg, of USP Doxorubicin Hydrochloride RS in each mL of the Standard preparation; P is the content, in µg per mg, of C27H29NO11·HCl in the USP Doxorubicin Hydrochloride RS; and rU and rS are the responses of the doxorubicin peak obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ahalya Wise, M.S.
Senior Scientific Liaison 1-301-816-8161 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2980
Pharmacopeial Forum: Volume No. 29(1) Page 66