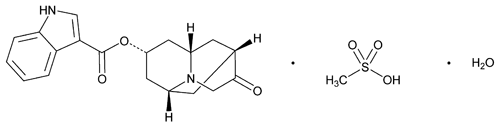
Dolasetron Mesylate
C19H20N2O3·CH4O3S·H2O 438.50
1H-Indole-3-carboxylic acid, octahydro-3-oxo-2,6-methano-2H-quinolizin-8-yl ester, (2 ,6
,6 ,8
,8 ,9a
,9a )-, monomethanesulfonate monohydrate;
)-, monomethanesulfonate monohydrate;
Indole-3-carboxylic acid, ester with (8r)-hexahydro-8-hydroxy-2,6-methano-2H-quinolizin-3(4H)-one, monomethanesulfonate monohydrate

 [115956-13-3].
[115956-13-3].
(doe las' e tron mes' i late).
C19H20N2O3·CH4O3S·H2O 438.50
1H-Indole-3-carboxylic acid, octahydro-3-oxo-2,6-methano-2H-quinolizin-8-yl ester, (2
Indole-3-carboxylic acid, ester with (8r)-hexahydro-8-hydroxy-2,6-methano-2H-quinolizin-3(4H)-one, monomethanesulfonate monohydrate
DEFINITION
Dolasetron Mesylate contains NLT 98.0% and NMT 102.0% of C19H20N2O3·CH4O3S·H2O, calculated on the as-is basis.
IDENTIFICATION
• B. Procedure
Sample solution:
1 mg/mL
Analysis:
Transfer 5–10 mg of 5,5'-methylenedisalicylic acid to a clean crucible, and heat in an oven at 150 for 5 min. Remove from the oven, and add 10 drops of the Sample solution. Return to the oven, and evaporate to dryness.
for 5 min. Remove from the oven, and add 10 drops of the Sample solution. Return to the oven, and evaporate to dryness.
Acceptance criteria:
A red or pink color (presence of methanesulfonic acid) develops in the white residue.
ASSAY
• Procedure
Mobile phase:
Acetonitrile, water, and 1 M ammonium formate (450:440:110), adding 0.19 mL of triethylamine to the acetonitrile portion
Standard solution:
0.04 mg/mL and 0.004 mg/mL respectively of USP Dolasetron Mesylate RS and indole-3-carboxylic acid in Mobile phase
Sample solution:
0.04 mg/mL of Dolasetron Mesylate in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 285 nm
Column:
4.6-mm × 15-cm; packing L1
Flow rate:
1 mL/min
Injection size:
20 µL
System suitability
Sample:
Standard solution
Suitability requirements
Resolution:
NLT 4 between indole-3-carboxylic acid and dolasetron mesylate
Tailing factor:
NMT 1.8
Relative standard deviation:
NMT 1.5% for replicate injections
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C19H20N2O3·CH4O3S·H2O in the Dolasetron Mesylate taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Dolasetron Mesylate RS in the Standard solution (mg/mL) |
| CU | = | = concentration of Dolasetron Mesylate in the Sample solution (mg/mL) |
Acceptance criteria:
98.0%–102.0% on the as-is basis
IMPURITIES
Organic Impurities
• Procedure
0.01 M Dibasic ammonium phosphate solution:
1.32 g/L of dibasic ammonium phosphate. Adjust with 2.0 M phosphoric acid to a pH of 7.0.
Diluent:
Acetonitrile and water (1:4)
Solution A:
Acetonitrile and 0.01 M Dibasic ammonium phosphate solution (53:1000), filtered and degassed
Solution B:
Acetonitrile and 0.01 M Dibasic ammonium phosphate solution (795:295), filtered and degassed
Mobile phase:
See the gradient table below.
| Time (min) |
Solution A (%) |
Solution B (%) |
|---|---|---|
| 0 | 100 | 0 |
| 28 | 0 | 100 |
| 38 | 0 | 100 |
| 40 | 100 | 0 |
| 50 | 100 | 0 |
System suitability solution:
0.004 mg/mL and 0.03 mg/mL, respectively, of indole and USP Dolasetron Mesylate RS in Diluent
Standard solution A:
0.03 mg/mL of USP Dolasetron Mesylate RS in Diluent
Standard solution B:
6 mg/mL and 0.0072 mg/mL, respectively, of USP Dolasetron Mesylate RS and USP Dolasetron Mesylate Related Compound A RS in Diluent
Sample solution:
6 mg/mL of Dolasetron Mesylate in Diluent
Chromatographic system
Mode:
LC
Detector:
UV 210 nm
Column:
4.6-mm × 25-cm; packing L7
Flow rate:
1.5 mL/min
Injection size:
100 µL
System suitability
Suitability requirements
Resolution:
NLT 1.5 between the first eluting peak, indole, and the second eluting peak, dolasetron mesylate from the System suitability solution. [Note—If the dolasetron mesylate peak is found to elute before the indole peak, condition the column as follows: fill up the column with Solution A, plug the column, and place the column in a convection oven at 105 for about 16 h. Retest the column. ]
for about 16 h. Retest the column. ]
Relative standard deviation:
NMT 5.0% for replicate injections of Standard solution A
Analysis
Samples:
Standard solution A, Standard solution B, and Sample solution
Calculate the percentage of dolasetron mesylate related compound A in the Dolasetron Mesylate taken:
Result = (rU/rS) × (CS/CU) × (Mr1/Mr2) × 100
| rU | = | = peak response of dolasetron mesylate related compound A from the Sample solution |
| rS | = | = peak response of dolasetron mesylate related compound A from the Standard solution B |
| CS | = | = concentration of USP Dolasetron Mesylate Related Compound A RS in the Standard solution B (mg/mL) |
| CU | = | = concentration of Dolasetron Mesylate in the Sample solution (mg/mL) |
| Mr1 | = | = molecular weight of dolasetron mesylate related compound A base, 181.2 |
| Mr2 | = | = molecular weight of dolasetron mesylate related compound A hydrochloride, 217.8 |
Calculate the percentage of each impurity (other than dolasetron mesylate related compound A) in the portion of Dolasetron Mesylate taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of each impurity from the Sample solution |
| rS | = | = peak response of dolasetron mesylate from the Standard solution A |
| CS | = | = concentration of USP Dolasetron Mesylate RS in the Standard solution A (mg/mL) |
| CU | = | = concentration of Dolasetron Mesylate in the Sample solution (mg/mL) |
Acceptance criteria
Individual impurities:
NMT 0.1%
Total impurities:
NMT 0.3%
[Note—The reporting level for impurities is 0.05%. ]
SPECIFIC TESTS
• Water Determination, Method Ia  921
921 :
Between 3.5% and 4.7%
:
Between 3.5% and 4.7%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well-closed containers, protected from light.
• USP Reference Standards  11
11
USP Dolasetron Mesylate Related Compound A RS
Hexahydro-8-hydroxy-2,6-methano-2H-quinolizin-3 (4H)-one, hydrochloride.
Hexahydro-8-hydroxy-2,6-methano-2H-quinolizin-3 (4H)-one, hydrochloride.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Elena Gonikberg, Ph.D.
Principal Scientific Liaison 1-301-816-8251 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2963
Pharmacopeial Forum: Volume No. 35(2) Page 272