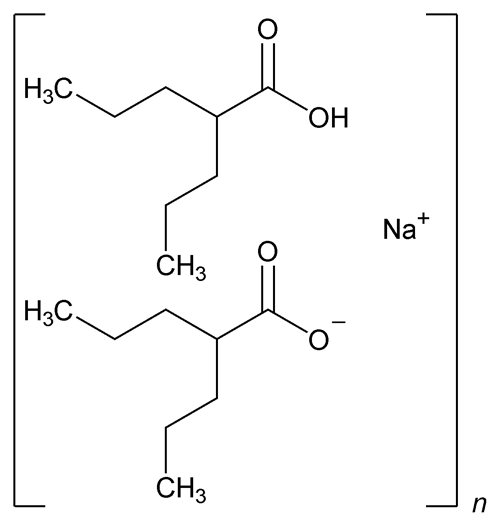
Divalproex Sodium
(C16H31NaO4)n 310.41
Pentanoic acid, 2-propyl-, sodium salt (2:1);
Sodium hydrogen bis(2-propylvalerate) oligomer.
(dye val' proe ex soe' dee um).
(C16H31NaO4)n 310.41
Pentanoic acid, 2-propyl-, sodium salt (2:1);
Sodium hydrogen bis(2-propylvalerate) oligomer.
DEFINITION
Divalproex Sodium contains NLT 98.0% and NMT 102.0% of available valproic acid (C8H16O2).
IDENTIFICATION
• B. Identification Tests—General, Sodium  191
191
Sample:
100 mg
Analysis:
Ignite the Sample.
Acceptance criteria:
Meets the requirements
ASSAY
• Procedure
Solution A:
3.5 g of monobasic sodium phosphate monohydrate in 900 mL of water. Adjust with phosphoric acid to a pH of 3.5. Dilute with water to 1 L.
Mobile phase:
Acetonitrile and Solution A (1:1)
Impurity stock solution:
0.5 mg/mL of USP Valproic Acid Related Compound A RS in acetonitrile
Standard stock solution:
5.0 mg/mL of USP Valproic Acid RS in Mobile phase
System suitability solution:
0.5 mg/mL of valproic acid from the Standard stock solution, and 5 µg/mL of valproic acid related compound A from the Impurity stock solution, in Mobile phase
Standard solution:
0.5 mg/mL of USP Valproic Acid RS from the Standard stock solution in Mobile phase
Sample solution:
0.5 mg/mL of Divalproex Sodium in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 215 nm
Column:
4.6-mm × 15-cm; 5-µm packing L7
Flow rate:
1 mL/min
Injection size:
20 µL
System suitability
Sample:
System suitability solution
[Note—The relative retention times for valproic acid related compound A and valproic acid are 0.69 and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 5.0 between valproic acid related compound A and valproic acid
Tailing factor:
NMT 1.5 for the valproic acid peak
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of available valproic acid (C8H16O2) in the portion of Divalproex Sodium taken:
Result = (rU/rS) × (CS/CU) × (Mr1/Mr2) × (1/F) × 100
| rU | = | = peak area from the Sample solution |
| rS | = | = peak area from the Standard solution |
| CS | = | = concentration of USP Valproic Acid RS in the Standard solution (mg/mL) |
| CU | = | = concentration of the Sample solution (mg/mL) |
| Mr1 | = | = molecular weight for divalproex sodium repeating unit, 310.41 |
| Mr2 | = | = molecular weight for valproic acid, 144.21 |
| F | = | = number of moles of valproic acid per mole of divalproex sodium repeating unit, 2 |
Acceptance criteria:
98.0%–102.0%
IMPURITIES
Inorganic Impurities
• Heavy Metals, Method II  231
231 :
NMT 20 ppm
:
NMT 20 ppm
SPECIFIC TESTS
• Water Determination, Method I  921
921 :
NMT 1.0%
:
NMT 1.0%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers. Store at room temperature.
• USP Reference Standards  11
11
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2945
Pharmacopeial Forum: Volume No. 36(5) Page 1178