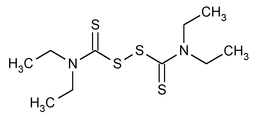
Disulfiram
C10H20N2S4 296.54
Thioperoxydicarbonic diamide [(H2N)C(S)]2S2, tetraethyl-;
Bis(diethylthiocarbamoyl) disulfide

 [97-77-8].
[97-77-8].
(dye sul' fi ram).
C10H20N2S4 296.54
Thioperoxydicarbonic diamide [(H2N)C(S)]2S2, tetraethyl-;
Bis(diethylthiocarbamoyl) disulfide
DEFINITION
Disulfiram contains NLT 98.0% and NMT 102.0% of C10H20N2S4.
IDENTIFICATION
• B.
The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Buffer:
6.8 g/L of monobasic potassium phosphate in water. Adjust with 45% potassium hydroxide solution to a pH of 7.0.
Mobile phase:
Methanol and Buffer (7:3)
Standard stock solution:
1 mg/mL of USP Disulfiram RS in alcohol. [Note—Discard this solution after 5 days. ]
Standard solution:
0.02 mg/mL of USP Disulfiram RS from the Standard stock solution diluted with Mobile phase. [Note—Prepare the Standard solution fresh daily. ]
Sample stock solution:
Transfer 50 mg of Disulfiram to a 50-mL volumetric flask, add 40 mL of alcohol, sonicate for 5 min to completely dissolve, cool, and dilute with alcohol to volume. [Note—Discard this solution after 5 days. ]
Sample solution:
0.02 mg/mL of Disulfiram from the Sample stock solution diluted with Mobile phase. [Note—Prepare the Sample solution fresh daily. ]
Chromatographic system
Mode:
LC
Detector:
UV 250 nm
Column:
4-mm × 15-cm; 5-µm packing L1
Flow rate:
1 mL/min
Injection size:
20 µL
System suitability
Sample:
Standard solution
Suitability requirements
Column efficiency:
NLT 1800 theoretical plates
Tailing factor:
NMT 2
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of C10H20N2S4 in the portion of Disulfiram taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Disulfiram RS in the Standard solution (µg/mL) |
| CU | = | = concentration of Disulfiram in the Sample solution (µg/mL) |
Acceptance criteria:
98.0%–102.0%
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.1%
:
NMT 0.1%
• Selenium  291
291 :
30 ppm
:
30 ppm
[Note—Perform this test only if selenium is a known inorganic process impurity. ]
SPECIFIC TESTS
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight, light-resistant containers.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2944
Pharmacopeial Forum: Volume No. 35(4) Page 848