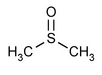
Dimethyl Sulfoxide
(dye meth' il sul fox' ide).
» Dimethyl Sulfoxide contains not less than 99.9 percent of C2H6OS, calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
A:
Infrared Absorption  197F
197F , potassium bromide plates being used.
, potassium bromide plates being used.
B:
Add 1.5 mL cautiously and dropwise to 2.5 mL of hydriodic acid in a test tube cooled in ice. Filter the mixture rapidly, and collect the precipitate. Dry the precipitate in vacuum: a deep violet, crystalline solid is obtained, and it is soluble in chloroform, yielding a red solution.
Specific gravity  841
841 :
between 1.095 and 1.101.
:
between 1.095 and 1.101.
Refractive index  831
831 :
between 1.4755 and 1.4775.
:
between 1.4755 and 1.4775.
Acidity—
Dissolve 50.0 g in 100 mL of water, and add phenolphthalein TS. If the solution remains colorless, titrate with 0.01 N sodium hydroxide until a pink color appears: not more than 5.0 mL of 0.01 N sodium hydroxide is consumed.
Water, Method I  921
921 :
not more than 0.1%. [note—Weigh and transfer the test specimen in an environment of low humidity to minimize absorption of atmospheric water. ]
:
not more than 0.1%. [note—Weigh and transfer the test specimen in an environment of low humidity to minimize absorption of atmospheric water. ]
Ultraviolet absorbance—
Maintain Dimethyl Sulfoxide in a water bath at a temperature of less than 20 [note—Do not freeze. ] , and purge with dry nitrogen for 30 minutes. Record the UV absorption spectrum between 270 and 350 nm in a 1-cm cell, using water as the blank: the spectrum is smooth with no absorption maxima; the absorbance at 275 nm is not more than 0.20, and the absorbance ratios, A285/A275 and A295/A275, at the wavelengths indicated by the subscripts, are not more than 0.65 and 0.45, respectively.
[note—Do not freeze. ] , and purge with dry nitrogen for 30 minutes. Record the UV absorption spectrum between 270 and 350 nm in a 1-cm cell, using water as the blank: the spectrum is smooth with no absorption maxima; the absorbance at 275 nm is not more than 0.20, and the absorbance ratios, A285/A275 and A295/A275, at the wavelengths indicated by the subscripts, are not more than 0.65 and 0.45, respectively.
Limit of nonvolatile residue—
Evaporate about 100.0 g to dryness in a tared, preconditioned dish on a hot plate under a suitable fume hood. Evaporate gently so boiling does not occur. The weight of the residue does not exceed 0.01%.
Related compounds—
System suitability solution
—Prepare a solution containing about 0.15 mg of dimethyl sulfone per mL and 0.1 mg of dibenzyl per mL in Dimethyl Sulfoxide.
Chromatographic system
(see Chromatography  621
621 )—The gas chromatograph is equipped with a flame-ionization detector and contains a 0.32-mm × 15-m column bonded with a 3-µm film of phase G2. The column temperature is programmed to change from 100
)—The gas chromatograph is equipped with a flame-ionization detector and contains a 0.32-mm × 15-m column bonded with a 3-µm film of phase G2. The column temperature is programmed to change from 100 to 170
to 170 at a rate of about 10
at a rate of about 10 per minute, the injection port is maintained at a temperature of about 210
per minute, the injection port is maintained at a temperature of about 210 , and the detector block is maintained at a temperature of about 220
, and the detector block is maintained at a temperature of about 220 . Helium is used as the carrier gas, flowing at a rate of 1.7 mL per minute, and the split ratio is 33:1. [note—The split ratio can be modified in order to optimize performance. ] Chromatograph the System suitability solution, and record the peak responses as directed for Procedure: the resolution, R, between dimethyl sulfone and dibenzyl is not less than 5.0; and the column efficiency, determined from the dimethyl sulfoxide peak, is not less than 1000 theoretical plates.
. Helium is used as the carrier gas, flowing at a rate of 1.7 mL per minute, and the split ratio is 33:1. [note—The split ratio can be modified in order to optimize performance. ] Chromatograph the System suitability solution, and record the peak responses as directed for Procedure: the resolution, R, between dimethyl sulfone and dibenzyl is not less than 5.0; and the column efficiency, determined from the dimethyl sulfoxide peak, is not less than 1000 theoretical plates.
Procedure—
Inject about 1 µL of Dimethyl Sulfoxide into the gas chromatograph, record the chromatograms, and measure the peak responses. Calculate the percentage of total impurities in the portion of Dimethyl Sulfoxide taken by the formula:
100(rU / rs)
in which rU is the sum of the areas of all the impurity peaks; and rs is the sum of the areas of all the peaks: not more than 0.1% of total impurities is found.
Assay—
Using the results from the test for Limit of nonvolatile residue and the test for Related compounds, calculate the percentage of C2H6OS in the portion of Dimethyl Sulfoxide taken by subtracting the percentage of nonvolatile residue and the percentage of total impurities found from 100.0%. [note—A correction for percent water is not applied to the result. ]
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Feiwen Mao, M.S.
Senior Scientific Liaison 1-301-816-8320 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2925
Pharmacopeial Forum: Volume No. 34(1) Page 88