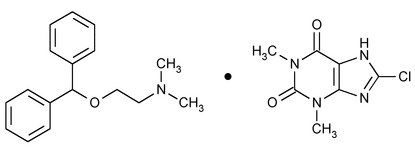Dimenhydrinate
(dye'' men hye' dri nate).
1H-Purine-2,6-dione, 8-chloro-3,7-dihydro-1,3-dimethyl-, compd. with 2-(diphenylmethoxy)-N,N-dimethylethanamine (1:1).
8-Chlorotheophylline, compound with 2-(diphenylmethoxy)-N,N-dimethylethylamine (1:1)
» Dimenhydrinate contains not less than 53.0 percent and not more than 55.5 percent of diphenhydramine (C17H21NO), and not less than 44.0 percent and not more than 47.0 percent of 8-chlorotheophylline (C7H7ClN4O2), calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
Identification—
Infrared Absorption  197K
197K .
.
Melting range  741
741 :
between 102
:
between 102 and 107
and 107 .
.
Loss on drying  731
731 —
Dry it in vacuum over phosphorus pentoxide for 24 hours: it loses not more than 0.5% of its weight.
—
Dry it in vacuum over phosphorus pentoxide for 24 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.3%.
:
not more than 0.3%.
Bromide and iodide—
Mix in a test tube 100 mg of Dimenhydrinate, 50 mg of sodium nitrite, and 10 mL of chloroform. Add 10 mL of 3 N hydrochloric acid, insert the stopper in the tube, and shake: the chloroform remains colorless.
Chloride—
When the ammoniacal filtrate from the precipitation of silver chlorotheophylline in the Assay for 8-chlorotheophylline is acidified preparatory to titration, the solution shows not more than a faint opalescence.
Assay for diphenhydramine—
Dissolve about 150 mg of Dimenhydrinate, accurately weighed, in 75 mL of glacial acetic acid, and titrate with 0.05 N perchloric acid VS, determining the endpoint potentiometrically. Perform a blank determination, and make any necessary correction. Each mL of 0.05 N perchloric acid is equivalent to 12.77 mg of diphenhydramine (C17H21NO).
Assay for 8-chlorotheophylline—
Place about 800 mg of Dimenhydrinate, accurately weighed, in a 200-mL volumetric flask, add 50 mL of water, 3 mL of 6 N ammonium hydroxide, and 6 mL of ammonium nitrate solution (1 in 10), and warm the mixture on a steam bath for 5 minutes. Add 25.0 mL of 0.1 N silver nitrate VS, mix, and warm on a steam bath for 15 minutes with frequent shaking. Cool, dilute with water to volume, mix, and allow the precipitate to settle. Filter through a dry filter paper, discarding the first 20 mL of the filtrate. Pipet 100 mL of the filtrate into a 250-mL flask, acidify with nitric acid, and add an excess of 3 mL of the acid. Add 2 mL of ferric ammonium sulfate TS, and titrate the excess silver nitrate with 0.1 N ammonium thiocyanate VS. Each mL of 0.1 N silver nitrate is equivalent to 21.46 mg of C7H7ClN4O2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Elena Gonikberg, Ph.D.
Principal Scientific Liaison 1-301-816-8251 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2922
Pharmacopeial Forum: Volume No. 33(5) Page 907