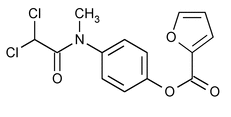
Diloxanide Furoate
(dye lox' a nide fure' oh ate).
» Diloxanide Furoate contains not less than 98.0 percent and not more than 102.0 percent of C14H11Cl2NO4, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers at controlled room temperature.
Identification—
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
14 µg per mL.
Medium:
alcohol. The solution exhibits a maximum at 258 nm.
C:
Burn 20 mg by employing the method under Oxygen-Flask Combustion  471
471 , using 10 mL of 1 N sodium hydroxide as the absorbing liquid. When the process is complete, acidify the liquid with nitric acid, and add a solution of silver nitrate (5 in 100). A white precipitate forms.
, using 10 mL of 1 N sodium hydroxide as the absorbing liquid. When the process is complete, acidify the liquid with nitric acid, and add a solution of silver nitrate (5 in 100). A white precipitate forms.
Melting range  741
741 :
between 114
:
between 114 and 116
and 116 .
.
Acidity—
Shake 3 g with 50 mL of water, filter, wash the residue with three 20-mL portions of water, and combine the filtrate and washings. Titrate with 0.1 N sodium hydroxide VS using phenolphthalein TS as the indicator: not more than 1.3 mL of 0.1 N sodium hydroxide is required for neutralization.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 to constant weight: it loses not more than 0.5%.
to constant weight: it loses not more than 0.5%.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Related compounds—
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture.
Test solution—
Dissolve an accurately weighed amount of Diloxanide Furoate in chloroform to obtain a solution having a concentration of about 100 mg per mL.
Standard solution—
Dilute 0.5 mL of Test solution with chloroform to 200 mL.
Application volume:
5 µL.
Developing solvent system:
a mixture of dichloromethane and methanol (96:4).
Procedure—
Proceed as directed for Thin-Layer Chromatography under Chromatography  621
621 . Examine the plate under short-wavelength UV light: any secondary spot obtained from the Test solution is not more intense than the spot obtained from the Standard solution.
. Examine the plate under short-wavelength UV light: any secondary spot obtained from the Test solution is not more intense than the spot obtained from the Standard solution.
Assay—
Dissolve about 300 mg of Diloxanide Furoate, accurately weighed, in 50.0 mL of dried pyridine, and titrate with 0.1 N tetrabutylammonium hydroxide VS, determining the endpoint potentiometrically using suitable electrodes (see Titrimetry  541
541 ). Perform a blank determination, and make any necessary correction. Each mL of 0.1 N tetrabutylammonium hydroxide is equivalent to 32.82 mg of C14H11Cl2NO4.
). Perform a blank determination, and make any necessary correction. Each mL of 0.1 N tetrabutylammonium hydroxide is equivalent to 32.82 mg of C14H11Cl2NO4.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Behnam Davani, Ph.D., M.B.A.
Senior Scientific Liaison 1-301-816-8394 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2915
Pharmacopeial Forum: Volume No. 27(1) Page 1778