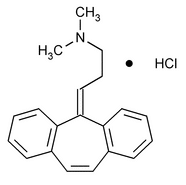
Cyclobenzaprine Hydrochloride
(sye'' kloe ben' za preen hye'' droe klor' ide).
1-Propanamine, 3-(5H-dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethyl-, hydrochloride.
N,N-Dimethyl-5H-dibenzo[a,d]cycloheptene-D5,
» Cyclobenzaprine Hydrochloride contains not less than 99.0 percent and not more than 101.0 percent of C20H21N·HCl, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
Identification—
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
15 µg per mL.
Medium:
methanol.
Absorptivities at 290 nm, calculated on the dried basis, do not differ by more than 3.0%.
C:
A solution (1 in 50) responds to the tests for Chloride  191
191 .
.
Melting range  741
741 :
between 215
:
between 215 and 219
and 219 , but the range between beginning and end of melting does not exceed 2
, but the range between beginning and end of melting does not exceed 2 .
.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 to constant weight: it loses not more than 1.0% of its weight.
to constant weight: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method II  231
231 :
0.001%.
:
0.001%.
Chromatographic purity—
Dissolve 100 mg in methanol, and dilute with methanol to 5.0 mL to obtain the Test solution. Dissolve a suitable quantity of USP Cyclobenzaprine Hydrochloride RS in methanol to obtain a Standard solution having a known concentration of about 20 mg per mL. Dilute a portion of this solution quantitatively and stepwise with methanol to obtain a Diluted standard solution having a concentration of about 100 µg per mL. Apply separate 5-µL portions of the three solutions to the starting line of a suitable thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel mixture and previously washed with methanol. Develop the chromatogram in a suitable chamber with a freshly prepared solvent system consisting of a mixture of acetone, toluene, and ammonium hydroxide (75:25:1) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber, air-dry, and view under short-wavelength UV light: the RF value of the principal spot from the Test solution corresponds to that of the Standard solution; and any other spot obtained from the Test solution does not exceed, in size or intensity, the principal spot obtained from the Diluted standard solution (0.5%).
) coated with a 0.25-mm layer of chromatographic silica gel mixture and previously washed with methanol. Develop the chromatogram in a suitable chamber with a freshly prepared solvent system consisting of a mixture of acetone, toluene, and ammonium hydroxide (75:25:1) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber, air-dry, and view under short-wavelength UV light: the RF value of the principal spot from the Test solution corresponds to that of the Standard solution; and any other spot obtained from the Test solution does not exceed, in size or intensity, the principal spot obtained from the Diluted standard solution (0.5%).
Assay—
Dissolve about 400 mg of Cyclobenzaprine Hydrochloride, accurately weighed, in about 80 mL of glacial acetic acid, add 15 mL of mercuric acetate TS, and titrate with 0.1 N perchloric acid VS, determining the endpoint potentiometrically, using a platinum ring electrode and a sleeve-type calomel electrode containing 0.1 N lithium perchlorate in glacial acetic acid (see Titrimetry  541
541 ). Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 31.19 mg of C20H21N·HCl.
). Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 31.19 mg of C20H21N·HCl.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2785
Pharmacopeial Forum: Volume No. 29(4) Page 1024