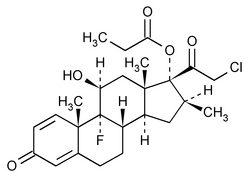
Clobetasol Propionate
C25H32ClFO5 466.97
Pregna-1,4-diene-3,20-dione, 21-chloro-9-fluoro-11-hydroxy-16-methyl-17-(1-oxopropoxy)-, (11 ,16
,16 )-;
)-;
21-Chloro-9-fluoro-11 ,17-dihydroxy-16
,17-dihydroxy-16 -methylpregna-1,4-diene-3,20-dione 17-propionate
-methylpregna-1,4-diene-3,20-dione 17-propionate


 [25122-46-7; 25122-41-2].
[25122-46-7; 25122-41-2].
(kloe bay' ta sol proe' pee oh nate).
C25H32ClFO5 466.97
Pregna-1,4-diene-3,20-dione, 21-chloro-9-fluoro-11-hydroxy-16-methyl-17-(1-oxopropoxy)-, (11
21-Chloro-9-fluoro-11
DEFINITION
Clobetasol Propionate contains NLT 97.0% and NMT 102.0% of C25H32ClFO5, calculated on the dried basis.
IDENTIFICATION
ASSAY
• Procedure
Solution A:
0.05 M monobasic sodium phosphate. Adjust with 85% phosphoric acid to a pH of 2.5.
Mobile phase:
Acetonitrile, methanol, and Solution A (19:4:17)
Internal standard solution:
0.2 mg/mL of beclomethasone dipropionate in methanol
Standard solution:
Dissolve a quantity of USP Clobetasol Propionate RS in methanol and Internal standard solution to obtain a final solution of 0.04 mg/mL of USP Clobetasol Propionate RS and 0.08 mg/mL of beclomethasone dipropionate.
System suitability solution:
0.001 mg/mL of USP Clobetasol Propionate Related Compound A RS and 0.1 mg/mL of USP Clobetasol Propionate RS in Mobile phase
Sample solution:
Transfer 4 mg of Clobetasol Propionate to a 100-mL volumetric flask, add 40.0 mL of Internal standard solution, and dilute with methanol to volume.
Chromatographic system
Mode:
LC
Detector:
UV 240 nm
Column:
4.6-mm × 15-cm; packing L1
Flow rate:
1 mL/min
Injection size:
10 µL
System suitability
Sample:
System suitability solution
[Note—The relative retention times for clobetasol propionate and clobetasol propionate related compound A are 1.0 and 1.1, respectively. ]
Suitability requirements
Resolution:
NLT 1.5 between clobetasol propionate and clobetasol propionate related compound A
Column efficiency:
NLT 5000 theoretical plates for the clobetasol peak
Tailing factor:
NMT 2.0 for the clobetasol peak
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
[Note—The relative retention times for clobetasol propionate and beclomethasone dipropionate are 1.0 and 1.6, respectively. ]
Calculate the percentage of C25H32ClFO5 in the portion of Clobetasol Propionate taken:
Result = (RU/RS) × (CS/CU) × 100
| RU | = | = ratio of the clobetasol propionate peak area to the internal standard peak area from the Sample solution |
| RS | = | = ratio of the clobetasol propionate peak area to the internal standard peak area from the Standard solution |
| CS | = | = concentration of USP Clobetasol Propionate RS in the Standard solution (mg/mL) |
| CU | = | = nominal concentration of clobetasol propionate in the Sample solution (mg/mL) |
Acceptance criteria:
97.0%–102.0% on the dried basis
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.1%, using a platinum crucible
:
NMT 0.1%, using a platinum crucible
• Heavy Metals, Method II  231
231 :
NMT 20 ppm
:
NMT 20 ppm
Organic Impurities
• Procedure
Solution A, Mobile phase, System suitability solution, and Chromatographic system:
Proceed as directed in the Assay.
Sample solution:
0.1 mg/mL of Clobetasol Propionate in Mobile phase
Analysis
Sample:
Sample solution
Calculate the percentage of each impurity in the portion of Clobetasol Propionate taken:
Result = (rU/rT) × 100
| rU | = | = peak area for each impurity |
| rT | = | = sum of the areas of all of the peaks |
Acceptance criteria
Any individual impurity:
NMT 1.0%
Total impurities:
NMT 2.5%
SPECIFIC TESTS
• Melting Range or Temperature  741
741 :
Approximately 196
:
Approximately 196
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 for 3 h: it loses NMT 2.0% of its weight.
for 3 h: it loses NMT 2.0% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight, light-resistant containers.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Domenick Vicchio, Ph.D.
Senior Scientific Liaison 1-301-998-6828 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2713
Pharmacopeial Forum: Volume No. 35(5) Page 1125