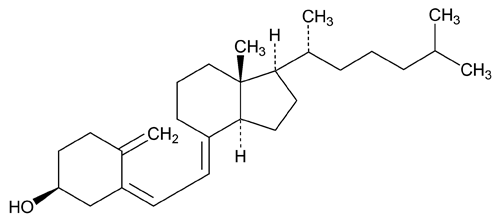
Cholecalciferol
(koe'' le kal sif' er ol).
DEFINITION
Cholecalciferol contains NLT 97.0% and NMT 103.0% of cholecalciferol (C27H44O).
IDENTIFICATION
• A. Infrared Absorption  197K
197K
Wavelength range:
2–12 µm
• B. Ultraviolet Absorption  197U
197U
Analytical wavelength:
265 nm
Sample solution:
10 µg/mL in alcohol
Acceptance criteria:
Meets the requirements in the chapter. Absorptivities do not differ by more than 3.0%.
• C.
Sample solution:
0.5 mg in 5 mL of chloroform
Analysis:
Add 0.3 mL of acetic anhydride and 0.1 mL of sulfuric acid to the Sample solution, and shake vigorously.
Acceptance criteria:
A bright red color is produced, and it rapidly changes through violet and blue to green.
• D. Thin-Layer Chromatography
[Note—For the Standard solution and the Sample solution, follow these procedures: use low-actinic glassware, dissolve the samples without heating, and use the solutions immediately. ]
Diluent:
10 mg/mL of squalane in chloroform
Standard solution:
50 mg/mL of USP Cholecalciferol RS in Diluent
Sample solution:
50 mg/mL of Cholecalciferol in Diluent
Chromatographic system
Mode:
TLC
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture
Application volume:
10 µL
Developing solvent system:
Cyclohexane and diethyl ether (1:1)
Spray reagent:
20 mg/mL of acetyl chloride in antimony trichloride TS
Analysis
Samples:
Standard solution and Sample solution
[Note—Perform the development and subsequent operations in the dark. ]
Place the plate in a chamber containing and equilibrated with Developing solvent system. Develop until the solvent front has moved about 15 cm above the line of application. Remove the plate, allow the solvent to evaporate, and spray with Spray reagent.
Acceptance criteria:
The Sample solution shows a yellowish-orange area (cholecalciferol) having the same RF value as the area of the Standard solution and may show below the cholecalciferol area a violet area, attributed to 7-dehydrocholesterol.
ASSAY
• Procedure
Dehydrated hexane:
Prepare a chromatographic column by packing a chromatographic tube, 8 × 60 cm, with 500 g of 50- to 250-µm chromatographic siliceous earth, activated by drying at 150 for 4 h. (See Chromatography
for 4 h. (See Chromatography  621
621 , Column Chromatography.) Pass 500 mL of hexane through the column, and collect the eluate in a glass-stoppered flask.
, Column Chromatography.) Pass 500 mL of hexane through the column, and collect the eluate in a glass-stoppered flask.
Mobile phase:
n-Amyl alcohol in Dehydrated hexane (3 in 1000)
System suitability solution:
250 mg of USP Vitamin D Assay System Suitability RS in 10 mL of a mixture of toluene and Mobile phase (1:1). Heat this solution, under reflux, at 90 for 45 min, and cool. [Note—This solution contains cholecalciferol, precholecalciferol, and trans-cholecalciferol. ]
for 45 min, and cool. [Note—This solution contains cholecalciferol, precholecalciferol, and trans-cholecalciferol. ]
[Note—For the stock solutions, follow these procedures: use low-actinic glassware, dissolve the samples without heating, and prepare the solutions fresh daily. ]
Standard stock solution:
0.6 mg/mL of USP Cholecalciferol RS in toluene
Standard solution:
120 µg/mL of USP Cholecalciferol RS in Mobile phase, prepared from Standard stock solution
Sample stock solution:
0.6 mg/mL of Cholecalciferol in toluene
Sample solution:
120 µg/mL of Cholecalciferol in Mobile phase, prepared from Sample stock solution
Chromatographic system
Mode:
LC
Detector:
UV 254 nm
Column:
4.6-mm × 25-cm; packing L3
Injection size:
5–10 µL
System suitability
Sample:
System suitability solution
[Note—The relative retention times for precholecalciferol, trans-cholecalciferol, and cholecalciferol are 0.4, 0.5, and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 1.0 between trans-cholecalciferol and precholecalciferol
Relative standard deviation:
NMT 2.0% for the peak response of cholecalciferol
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of cholecalciferol (C27H44O) in the portion of Cholecalciferol taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response from the Sample solution |
| rS | = | = peak response from the Standard solution |
| CS | = | = concentration of USP Cholecalciferol RS in the Standard solution (µg/mL) |
| CU | = | = concentration of Cholecalciferol in the Sample solution (µg/mL) |
Acceptance criteria:
97.0%–103.0%
SPECIFIC TESTS
• Optical Rotation, Specific Rotation  781S
781S
Sample solution:
5 mg/mL in alcohol. [Note—Prepare and use the solution without delay. Use Cholecalciferol from a container opened not longer than 30 min. ]
Acceptance criteria:
+105 to +112
to +112
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in hermetically sealed containers under nitrogen, and store in a cool place protected from light.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Huy T. Dinh, M.S.
Scientific Liaison 1-301-816-8594 |
(DS2010) Monographs - Dietary Supplements |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2652