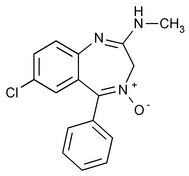
Chlordiazepoxide
(klor'' dye az'' e pox' ide).
3H-1,4-Benzodiazepin-2-amine, 7-chloro-N-methyl-5-phenyl-, 4-oxide.
7-Chloro-2-(methylamino)-5-phenyl-3H-1,4-benzodiazepine 4-oxide
» Chlordiazepoxide contains not less than 98.0 percent and not more than 102.0 percent of C16H14ClN3O, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation as obtained in the Assay.
C:
To about 20 mg add 5 mL of hydrochloric acid and 10 mL of water, and heat to boiling to effect hydrolysis. To the cooled solution add 2 mL of sodium nitrite solution (1 in 1000), shake, add 1 mL of ammonium sulfamate solution (1 in 200), then shake for 2 minutes, and add 1 mL of N-(1-naphthyl)ethylenediamine dihydrochloride solution (1 in 1000): a reddish violet color is produced.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 3 hours: it loses not more than 0.3% of its weight.
for 3 hours: it loses not more than 0.3% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method II  231
231 :
0.002%.
:
0.002%.
Related compounds—
Transfer 50.0 mg to a 10-mL conical flask, add 2.5 mL of acetone, and shake. Allow any undissolved particles to settle, and apply 50 µL of the supernatant to a suitable thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel. Apply to the same plate 10 µL of an acetone solution containing 100 µg per mL of USP Chlordiazepoxide Related Compound A RS and 10 µL of an acetone solution containing 10 µg per mL of USP 2-Amino-5-chlorobenzophenone RS. Develop the chromatogram in a chromatographic chamber (not previously saturated with the developing solvent) in ethyl acetate until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow the solvent to evaporate. Locate the spots on the plate by lightly spraying with 2 N sulfuric acid, drying at 105
) coated with a 0.25-mm layer of chromatographic silica gel. Apply to the same plate 10 µL of an acetone solution containing 100 µg per mL of USP Chlordiazepoxide Related Compound A RS and 10 µL of an acetone solution containing 10 µg per mL of USP 2-Amino-5-chlorobenzophenone RS. Develop the chromatogram in a chromatographic chamber (not previously saturated with the developing solvent) in ethyl acetate until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow the solvent to evaporate. Locate the spots on the plate by lightly spraying with 2 N sulfuric acid, drying at 105 for 15 minutes, and then spraying in succession with sodium nitrite solution (1 in 1000), ammonium sulfamate solution (1 in 200), and N-(1-naphthyl)ethylenediamine dihydrochloride solution (1 in 1000). Any spots from the test solution are not greater in size or intensity than the spots at the respective RF values produced by the Standard solutions, corresponding to not more than 0.1% of chlordiazepoxide related compound A, and to not more than 0.01% of 2-amino-5-chlorobenzophenone.
for 15 minutes, and then spraying in succession with sodium nitrite solution (1 in 1000), ammonium sulfamate solution (1 in 200), and N-(1-naphthyl)ethylenediamine dihydrochloride solution (1 in 1000). Any spots from the test solution are not greater in size or intensity than the spots at the respective RF values produced by the Standard solutions, corresponding to not more than 0.1% of chlordiazepoxide related compound A, and to not more than 0.01% of 2-amino-5-chlorobenzophenone.
Assay—
[note—Use low-actinic glassware in this procedure. ]
Mobile phase—
Prepare a filtered and degassed mixture of methanol and water (60:40). Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Standard preparation—
Dissolve an accurately weighed quantity of USP Chlordiazepoxide RS in Mobile phase, and dilute quantitatively, and stepwise if necessary, with Mobile phase to obtain a solution having a known concentration of about 200 µg per mL.
Assay preparation—
Transfer about 100 mg of Chlordiazepoxide, accurately weighed, to a 50-mL volumetric flask, dissolve in Mobile phase, sonicate for 5 minutes, dilute with Mobile phase to volume, and mix. Pass this solution through a membrane filter having a 0.5-µm or finer porosity. Transfer 10 mL of this solution to a 100-mL volumetric flask, dilute with Mobile phase, and mix.
Chromatographic system
(see Chromatography  621
621 )—The liquid chromatograph is equipped with a 254-nm detector and a 3.9-mm × 30-cm column that contains packing L1. The flow rate is about 1 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the column efficiency is not less than 3600 theoretical plates: the tailing factor is not more than 2.0; and the relative standard deviation for replicate injections is not more than 2.0%.
)—The liquid chromatograph is equipped with a 254-nm detector and a 3.9-mm × 30-cm column that contains packing L1. The flow rate is about 1 mL per minute. Chromatograph the Standard preparation, and record the peak responses as directed for Procedure: the column efficiency is not less than 3600 theoretical plates: the tailing factor is not more than 2.0; and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 5 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of C16H14ClN3O in the portion of Chlordiazepoxide taken by the formula:
0.5C(rU / rS)
in which C is the concentration, in µg per mL, of the USP Reference Standard in the Standard preparation; and rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2616