Microcrystalline Cellulose
Cellulose

 [9004-34-6].
[9004-34-6].
Cellulose
DEFINITION
Microcrystalline Cellulose is purified, partially depolymerized cellulose prepared by treating alpha cellulose, obtained as a pulp from fibrous plant material, with mineral acids.
IDENTIFICATION
• A. Procedure
Iodinated zinc chloride solution:
Dissolve 20 g of zinc chloride and 6.5 g of potassium iodide in 10.5 mL of water. Add 0.5 g of iodine, and shake for 15 min.
Sample:
10 mg
Analysis:
Place the Sample on a watch glass, and disperse in 2 mL of Iodinated zinc chloride solution.
Acceptance criteria:
The substance takes on a violet-blue color.
• B. Procedure
Sample:
1.3 g of Microcrystalline Cellulose, accurately weighed to 0.1 mg
Analysis:
Transfer the Sample to a 125-mL conical flask. Add 25.0 mL of water and 25.0 mL of 1.0 M cupriethylenediamine hydroxide solution. Immediately purge the solution with nitrogen, insert the stopper, and shake on a wrist-action shaker, or other suitable mechanical shaker, until completely dissolved. Transfer an appropriate volume of the Sample solution to a calibrated number 150 Cannon-Fenske, or equivalent, viscometer. Allow the solution to equilibrate at 25 ± 0.1 for NLT 5 min. Time the flow between the two marks on the viscometer, and record the flow time, t1, in s.
for NLT 5 min. Time the flow between the two marks on the viscometer, and record the flow time, t1, in s.
Calculate the kinematic viscosity, (KV)1, of the Microcrystalline Cellulose taken:
Result = t1 × k1
| t1 | = | = flow time (s) |
| k1 | = | = viscometer constant (see Viscosity |
Obtain the flow time, t2, for 0.5 M cupriethylenediamine hydroxide solutions using a number 100 Cannon-Fenske, or equivalent, viscometer.
Calculate the kinematic viscosity, (KV)2, of the solvent:
Result = t2 × k2
| t2 | = | = flow time for 0.5 M cupriethylenediamine hydroxide solutions (s) |
| k2 | = | = viscometer constant |
Determine the relative viscosity, 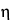 rel, of the Microcrystalline Cellulose specimen taken:
rel, of the Microcrystalline Cellulose specimen taken:
Result = (KV)1/(KV)2
| (KV)1 | = | = kinematic viscosity of the Microcrystalline Cellulose taken |
| (KV)2 | = | = kinematic viscosity of the solvent |
Determine the intrinsic viscosity, [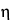 ]c, by interpolation, using the Intrinsic Viscosity Table in the Reference Tables section.
]c, by interpolation, using the Intrinsic Viscosity Table in the Reference Tables section.
Calculate the degree of polymerization, P:
Result = (95) × [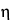 ]c/WS × [(100
]c/WS × [(100  %LOD)/100]
%LOD)/100]
| [ | = | = intrinsic viscosity |
| WS | = | = weight of the Microcrystalline Cellulose taken (g) |
| %LOD | = | = value obtained from the test for Loss on Drying |
Acceptance criteria:
The degree of polymerization is not greater than 350.
IMPURITIES
SPECIFIC TESTS
• Microbial Enumeration Tests  61
61 and Tests for Specified Microorganisms
and Tests for Specified Microorganisms  62
62 :
The total aerobic microbial count does not exceed 1000 cfu/g, and the total combined molds and yeasts count does not exceed 100 cfu/g. It meets the requirements of the tests for absence of Staphylococcus aureus and Pseudomonas aeruginosa and for the absence of Escherichia coli and Salmonella species.
:
The total aerobic microbial count does not exceed 1000 cfu/g, and the total combined molds and yeasts count does not exceed 100 cfu/g. It meets the requirements of the tests for absence of Staphylococcus aureus and Pseudomonas aeruginosa and for the absence of Escherichia coli and Salmonella species.
• Conductivity
Sample:
5 g
Analysis:
Shake the Sample with 40 mL of water for 20 min, and centrifuge. Retain the supernatant for use in the pH test. Using an appropriate conductivity meter that has been standardized with a potassium chloride conductivity calibration standard having a conductivity of 100 µS/cm, measure the conductivity of the supernatant after a stable reading is obtained, and measure the conductivity of the water used to prepare the test specimen.
Acceptance criteria:
The conductivity of the supernatant does not exceed the conductivity of the water by more than 75 µS/cm.
• pH  791
791 :
5.0–7.5 in the supernatant obtained in the Conductivity test
:
5.0–7.5 in the supernatant obtained in the Conductivity test
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 for 3 h: it loses NMT 7.0% of its weight, or some other lower percentage, or is within a percentage range, as specified in the labeling.
for 3 h: it loses NMT 7.0% of its weight, or some other lower percentage, or is within a percentage range, as specified in the labeling.
• Bulk Density
Analysis:
Use a volumeter that has been fitted with a 10-mesh screen. The volumeter is freestanding of the brass or stainless steel cup, which is calibrated to a capacity of 25.0 ± 0.05 mL and has an inside diameter of 30.0 ± 2.0 mm. Weigh the empty cup, position it under the chute, and slowly pour the powder from a height of 5.1 cm (2 in) above the funnel through the volumeter, at a rate suitable to prevent clogging, until the cup overflows. [Note—If excessive clogging of the screen occurs, remove the screen. ] Level the excess powder, and weigh the filled cup. Calculate the bulk density by dividing the weight of the powder in the cup by the volume of the cup.
Acceptance criteria:
The bulk density is within the labeled specification.
• Particle Size Distribution
[Note—In cases where there are no functionality-related concerns regarding the particle size distribution of the article, this test may be omitted. ]
Where the labeling states the particle size distribution, determine the particle size distribution as directed in Particle Size Distribution Estimation by Analytical Sieving  786
786 , or by a suitable validated procedure.
, or by a suitable validated procedure.
• Water-Soluble Substances
Sample:
5.0 g
Analysis:
Shake the Sample with 80 mL of water for 10 min, and pass with the aid of a vacuum through filter paper (Whatman No. 42 or equivalent) into a vacuum flask. Transfer the filtrate to a tared beaker, evaporate to dryness without charring, dry at 105 for 1 h, cool in a desiccator, and weigh.
for 1 h, cool in a desiccator, and weigh.
Acceptance criteria:
The difference between the weight of the residue and the weight obtained from a blank determination does not exceed 12.5 mg (0.25%).
• Ether-Soluble Substances
Sample:
10.0 g
Analysis:
Place the Sample in a chromatographic column having an internal diameter of about 20 mm, and pass 50 mL of peroxide-free ether through the column. Evaporate the eluate to dryness in a previously dried and tared evaporating dish with the aid of a current of air in a fume hood. After all the ether has evaporated, dry the residue at 105 for 30 min, cool in a desiccator, and weigh.
for 30 min, cool in a desiccator, and weigh.
Acceptance criteria:
The difference between the weight of the residue and the weight obtained from a blank determination does not exceed 5.0 mg (0.05%).
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight containers.
• Labeling:
The labeling indicates the nominal loss on drying, bulk density, and degree of polymerization values. Degree of polymerization compliance is determined using Identification test B. Where the particle size distribution is stated in the labeling, proceed as directed in the test for Particle Size Distribution. The labeling indicates with which technique the particle size distribution was determined if a technique other than analytical sieving was used; and the labeling indicates the d10, d50, and d90 values and the range for each.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology | |
| Radhakrishna S Tirumalai, Ph.D.
Principal Scientific Liaison 1-301-816-8339 |
(GCM2010) General Chapters - Microbiology |
USP35–NF30 Page 1746
Pharmacopeial Forum: Volume No. 31(5) Page 1421