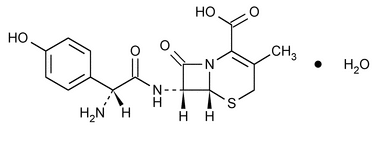
Cefadroxil
(sef'' a drox' il).
5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, 7-[[amino(4-hydroxyphenyl)acetyl]amino]-3-methyl-8-oxo-, monohydrate [6R-[6
(6R,7R)-7-[(R)-2-Amino-2-(p-hydroxyphenyl)acetamido]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid monohydrate
Hemihydrate 372.39
Anhydrous 363.40
» Cefadroxil has a potency equivalent to not less than 950 µg and not more than 1050 µg of C16H17N3O5S per mg, calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight containers.
Labeling—
The hemihydrate form is so labeled.
Identification—
B:
Place a suitable thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of binder-free silica gel in a chamber containing a mixture of n-hexane and tetradecane (95:5) to a depth of about 1 cm, allow the solvent front to move the length of the plate, remove the plate from the chamber, and allow the solvent to evaporate. Apply 20 µL each of a solution of Cefadroxil in water containing 2 mg per mL and a similarly prepared Standard solution of USP Cefadroxil RS to this plate. Allow the spots to dry, and develop the chromatogram in a solvent system consisting of 0.1 M citric acid, 0.1 M dibasic sodium phosphate, and a 1 in 15 solution of ninhydrin in acetone (60:40:1.5) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow to air-dry. Spray the plate with a 1 in 500 solution of ninhydrin in dehydrated alcohol [note—Protect this solution from light ] , dry for 10 minutes at 110
) coated with a 0.25-mm layer of binder-free silica gel in a chamber containing a mixture of n-hexane and tetradecane (95:5) to a depth of about 1 cm, allow the solvent front to move the length of the plate, remove the plate from the chamber, and allow the solvent to evaporate. Apply 20 µL each of a solution of Cefadroxil in water containing 2 mg per mL and a similarly prepared Standard solution of USP Cefadroxil RS to this plate. Allow the spots to dry, and develop the chromatogram in a solvent system consisting of 0.1 M citric acid, 0.1 M dibasic sodium phosphate, and a 1 in 15 solution of ninhydrin in acetone (60:40:1.5) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow to air-dry. Spray the plate with a 1 in 500 solution of ninhydrin in dehydrated alcohol [note—Protect this solution from light ] , dry for 10 minutes at 110 , and examine the chromatogram: the RF value of the principal spot obtained from the test solution corresponds to that obtained from the Standard solution.
, and examine the chromatogram: the RF value of the principal spot obtained from the test solution corresponds to that obtained from the Standard solution.
Crystallinity  695
695 :
meets the requirements.
:
meets the requirements.
pH  791
791 :
between 4.0 and 6.0, in a suspension containing 50 mg per mL.
:
between 4.0 and 6.0, in a suspension containing 50 mg per mL.
Water, Method I  921
921 :
between 4.2% and 6.0%, except that where it is labeled as being in the hemihydrate form it is between 2.4% and 4.5%.
:
between 4.2% and 6.0%, except that where it is labeled as being in the hemihydrate form it is between 2.4% and 4.5%.
Chromatographic purity—
Adsorbent:
a 0.25-mm layer of chromatographic silica gel mixture.
Solvent—
Prepare a mixture of alcohol, water, and 2.4 N hydrochloric acid (75:22:3).
Test solution—
Prepare a solution of Cefadroxil in Solvent containing 25 mg per mL.
Standard solution 1—
Dilute 1.0 mL of the Test solution with Solvent to 100 mL, and mix.
Standard solution 2—
Prepare a solution in Solvent containing 0.25 mg each of 7-aminodesacetoxycephalosporanic acid and d- -4-hydroxyphenylglycine per mL.
-4-hydroxyphenylglycine per mL.
Standard solution 3—
Prepare a solution in Solvent containing 0.25 mg of d- -4-hydroxyphenylglycine per mL.
-4-hydroxyphenylglycine per mL.
Resolution solution—
Mix 1.0 mL of the Test solution and 1.0 mL of Standard solution 2.
Developing solvent system:
a mixture of ethyl acetate, alcohol, water, and formic acid (14:5:5:1).
Procedure—
Apply separate 2-µL portions of the Test solution, Standard solution 1, Standard solution 2, and Standard solution 3, and a 4-µL portion of the Resolution solution to a suitable thin-layer chromatographic plate (see Thin-Layer Chromatography under Chromatography  621
621 ), and develop the chromatograms until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, allow the plate to dry, and examine the chromatograms under short-wavelength UV light: any secondary spot in the chromatogram obtained from the Test solution corresponding to 7-aminodesacetoxycephalosporanic acid or d-
), and develop the chromatograms until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, allow the plate to dry, and examine the chromatograms under short-wavelength UV light: any secondary spot in the chromatogram obtained from the Test solution corresponding to 7-aminodesacetoxycephalosporanic acid or d- -4-hydroxyphenylglycine is not more intense than the corresponding spot in the chromatogram obtained from Standard solution 2 (1.0%); and any spot, other than the principal spot and any spot corresponding to 7-aminodesacetoxycephalosporanic acid or d-
-4-hydroxyphenylglycine is not more intense than the corresponding spot in the chromatogram obtained from Standard solution 2 (1.0%); and any spot, other than the principal spot and any spot corresponding to 7-aminodesacetoxycephalosporanic acid or d- -4-hydroxyphenylglycine, is not more intense than the principal spot in the chromatogram obtained from Standard solution 1 (1.0%). In a valid test, the chromatogram obtained from the Resolution solution shows three clearly separated spots.
-4-hydroxyphenylglycine, is not more intense than the principal spot in the chromatogram obtained from Standard solution 1 (1.0%). In a valid test, the chromatogram obtained from the Resolution solution shows three clearly separated spots.
Dimethylaniline  223
223 :
meets the requirement.
:
meets the requirement.
Assay—
pH 5.0 Buffer—
Dissolve 13.6 g of monobasic potassium phosphate in water to make 2000 mL of solution. Adjust with 10 N potassium hydroxide to a pH of 5.0, and mix.
Mobile phase—
Prepare a suitable mixture of pH 5.0 Buffer and acetonitrile (960:40), and pass through a filter having a 0.5-µm or finer porosity. Make adjustments if necessary (see System Suitability under Chromatography  621
621 ). Increasing the acetonitrile content of the Mobile phase decreases the retention time of cefadroxil, and decreasing the acetonitrile content increases the retention time.
). Increasing the acetonitrile content of the Mobile phase decreases the retention time of cefadroxil, and decreasing the acetonitrile content increases the retention time.
Standard preparation—
Dissolve an accurately weighed quantity of USP Cefadroxil RS in pH 5.0 Buffer to obtain a solution having a known concentration of about 1.06 mg per mL. This solution contains the equivalent of about 1000 µg of cefadroxil (C16H17N3O5S) per mL. Use this solution on the day prepared.
Assay preparation—
Transfer about 212 mg of Cefadroxil, accurately weighed, to a 200-mL volumetric flask, dilute with pH 5.0 Buffer to volume, and stir by mechanical means for 5 minutes until dissolved. Use this solution on the day prepared.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 230-nm detector and a 4-mm × 25-cm column that contains packing L1. The flow rate is about 1.5 mL per minute. Chromatograph the Standard preparation, and record the responses as directed for Procedure: the capacity factor, k ¢, is between 2.0 and 3.5; the column efficiency determined from the analyte peak is not less than 1800 theoretical plates; the tailing factor for the analyte peak is not more than 2.2; and the relative standard deviation for replicate injections is not more than 2.0%.
)—
The liquid chromatograph is equipped with a 230-nm detector and a 4-mm × 25-cm column that contains packing L1. The flow rate is about 1.5 mL per minute. Chromatograph the Standard preparation, and record the responses as directed for Procedure: the capacity factor, k ¢, is between 2.0 and 3.5; the column efficiency determined from the analyte peak is not less than 1800 theoretical plates; the tailing factor for the analyte peak is not more than 2.2; and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 10 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the areas for the major peaks. Calculate the quantity, in µg, of cefadroxil (C16H17N3O5S) in each mg of the Cefadroxil taken by the formula:
200(CE/W)(rU / rS)
in which C is the concentration, in mg per mL, of USP Cefadroxil RS taken to prepare the Standard preparation; E is the cefadroxil equivalent, in µg per mg, of USP Cefadroxil RS; W is weight, in mg, of the portion of Cefadroxil taken; and rU and rS are the cefadroxil peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ahalya Wise, M.S.
Senior Scientific Liaison 1-301-816-8161 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2529
Pharmacopeial Forum: Volume No. 29(5) Page 1436