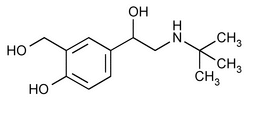
Albuterol
(al bue' ter ol).
1,3-Benzenedimethanol,
» Albuterol contains not less than 98.5 percent and not more than 101.0 percent of C13H21NO3, calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers.
Identification—
Solution:
80 µg per mL.
Medium:
0.1 N hydrochloric acid.
Water, Method I  921
921 :
not more than 0.5%.
:
not more than 0.5%.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Chromatographic purity—
Standard solution—
Dissolve an accurately weighed quantity of USP Albuterol RS in methanol to obtain a solution having a known concentration of 0.10 mg per mL.
Test solution—
Dissolve an accurately weighed quantity of Albuterol in methanol to obtain a solution having a concentration of 20 mg per mL.
Procedure—
Apply 10-µL aliquots of the Test and the Standard solution to separate points on a thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel. Place the plate in a saturated chromatographic chamber, and develop the chromatogram in a solvent system consisting of a mixture of methyl isobutyl ketone, isopropyl alcohol, ethyl acetate, water, and ammonium hydroxide (50:45:35:18:3) until the solvent front has moved three-fourths of the length of the plate. Remove the plate from the developing chamber, air-dry, and expose it to iodine vapor: any spot, other than the principal spot, obtained from the Test solution is not greater in size and intensity than the spot produced by the Standard solution (0.5%), and the sum of the impurities is not greater than 2.0%.
) coated with a 0.25-mm layer of chromatographic silica gel. Place the plate in a saturated chromatographic chamber, and develop the chromatogram in a solvent system consisting of a mixture of methyl isobutyl ketone, isopropyl alcohol, ethyl acetate, water, and ammonium hydroxide (50:45:35:18:3) until the solvent front has moved three-fourths of the length of the plate. Remove the plate from the developing chamber, air-dry, and expose it to iodine vapor: any spot, other than the principal spot, obtained from the Test solution is not greater in size and intensity than the spot produced by the Standard solution (0.5%), and the sum of the impurities is not greater than 2.0%.
Assay—
Dissolve about 400 mg of Albuterol, accurately weighed, in 50 mL of glacial acetic acid, add 2 drops of crystal violet TS, and titrate with 0.1 N perchloric acid VS. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 23.93 mg of C13H21NO3.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2084
Pharmacopeial Forum: Volume No. 27(3) Page 2505