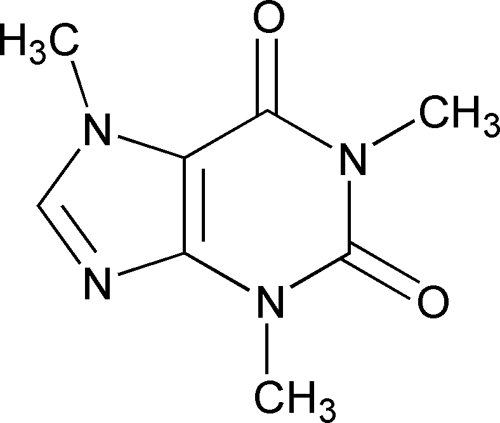
Caffeine
C8H10N4O2·H2O 212.21
C8H10N4O2 194.19
1H-Purine-2,6-dione, 3,7-dihydro-1,3,7-trimethyl-;
1,3,7-Trimethylxanthine

 [58-08-2].
[58-08-2].
Monohydrate

 [5743-12-4].
[5743-12-4].
(kaf' een).
C8H10N4O2·H2O 212.21
C8H10N4O2 194.19
1H-Purine-2,6-dione, 3,7-dihydro-1,3,7-trimethyl-;
1,3,7-Trimethylxanthine
Monohydrate
DEFINITION
Caffeine is anhydrous or contains one molecule of water of hydration. It contains NLT 98.5% and NMT 101.0% of C8H10N4O2, calculated on the anhydrous basis.
IDENTIFICATION
• B.
The retention time of the caffeine peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Buffer:
0.82 g/L of anhydrous sodium acetate
Mobile phase:
Acetonitrile, tetrahydrofuran, and Buffer (25:20:955). Adjust with glacial acetic acid to a pH of 4.5.
System suitability solution:
0.02 mg/mL of theophylline in Mobile phase. Shake, and sonicate if necessary, to dissolve.
Standard solution:
Transfer 5.0 mg of USP Caffeine RS to a 25-mL volumetric flask. Add 5.0 mL of the System suitability solution and 10 mL of Mobile phase. Shake, and sonicate if necessary. Dilute with Mobile phase to volume, and filter.
Sample solution:
0.2 mg/mL of Caffeine in Mobile phase. [Note—Shake, and sonicate if necessary, to dissolve. ]
Chromatographic system
Mode:
LC
Detector:
UV 275 nm
Column:
4.6-mm × 15-cm; packing L1
Flow rate:
1 mL/min
Injection size:
10 µL
System suitability
Sample:
Standard solution
[Note—The relative retention times for theophylline and caffeine are 0.69 and 1.0, respectively. ]
Suitability requirements
Resolution:
NLT 6.0 between theophylline and caffeine
Tailing factor:
NMT 2.0 for theophylline and caffeine peaks
Relative standard deviation:
NMT 2.0%
Analysis
Samples:
Standard solution and Sample solution
Calculate the percentage of caffeine (C8H10N4O2) in the portion of Caffeine taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak response of caffeine from the Sample solution |
| rS | = | = peak response of caffeine from the Standard solution |
| CS | = | = concentration of USP Caffeine RS in the Standard solution (mg/mL) |
| CU | = | = nominal concentration of Caffeine in the Sample solution (mg/mL) |
Acceptance criteria:
98.5%–101.0% on the anhydrous basis
IMPURITIES
• Residue on Ignition  281
281 :
NMT 0.1%
:
NMT 0.1%
• Heavy Metals, Method II  231
231 :
NMT 10 ppm
:
NMT 10 ppm
• Organic Impurities
Mobile phase, Standard solution, Sample solution, Chromatographic system, and System suitability:
Proceed as directed in the Assay.
Analysis
Sample:
Sample solution
Calculate the percentage of each impurity in the portion of Caffeine taken:
Result = (rU/rT) × 100
| rU | = | = peak response for each impurity from the Sample solution |
| rT | = | = sum of the responses of all the peaks from the Sample solution |
Acceptance criteria
Individual impurities:
NMT 0.1%
Total impurities:
NMT 0.1%
SPECIFIC TESTS
• Water Determination, Method III  921
921 :
Dry a sample at 80
:
Dry a sample at 80 for 4 h: the anhydrous form loses NMT 0.5% of its weight, and the hydrous form loses NMT 8.5% of its weight.
for 4 h: the anhydrous form loses NMT 0.5% of its weight, and the hydrous form loses NMT 8.5% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve hydrous Caffeine in tight containers. Preserve anhydrous Caffeine in well-closed containers.
• Labeling:
Label it to indicate whether it is anhydrous or hydrous.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2427
Pharmacopeial Forum: Volume No. 34(5) Page 1150