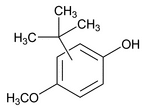
Butylated Hydroxyanisole
(bue' ti lay'' ted hye drox'' ee an' i sole).
DEFINITION
Butylated Hydroxyanisole contains NLT 98.5% of C11H16O2.
IDENTIFICATION
• A. Procedure
Analysis:
To 5 mL of 0.1 mg/mL solution in 72% alcohol add 2 mL of 20 mg/mL sodium borate solution and 1 mL of 0.1 mg/mL solution of 2,6-dichloroquinone-chlorimide in dehydrated alcohol, and mix.
Acceptance criteria:
A blue color is produced.
B.
The retention times of 3-tert-butyl-4-hydroxyanisole and 2-tert-butyl-4-hydroxyanisole from the Sample solution correspond to those from the Standard solution, as obtained in the Assay.
ASSAY
• Procedure
Solution A:
5% acetic acid
Mobile phase:
Acetonitrile and Solution A (45:55)
Standard solution:
90 µg/mL of USP 3-tert-Butyl-4-hydroxyanisole RS and 10 µg/mL of USP 2-tert-Butyl-4-hydroxyanisole RS in Mobile phase
Sample solution:
100 µg/mL of Butylated Hydroxyanisole in Mobile phase
Chromatographic system
Mode:
LC
Detector:
UV 290 nm
Column:
4.6-mm × 75-mm; 3.5-µm packing L1
Column temperature:
30
Flow rate:
1.2 mL/min
Injection size:
20 µL
System suitability
Sample:
Standard solution
[Note—The retention times of 2-tert-butyl-4-hydroxyanisole and 3-tert-butyl-4-hydroxyanisole are about 4.2 and 4.6 min, respectively. ]
Suitability requirements
Resolution:
NLT 1.5 between the 3-tert-butyl-4-hydroxyanisole isomer and 2-tert-butyl-4-hydroxyanisole isomer peaks
Tailing factor:
NMT 1.5
Relative standard deviation:
NMT 2.0% for the 3-tert-butyl-4-hydroxyanisole isomer and 2-tert-butyl-4-hydroxyanisole isomer peaks
Analysis
Samples:
Standard solution and Sample solution
Measure the peak areas for each isomer. Calculate the percentage of each isomer in the portion of Butylated Hydroxyanisole taken:
Result = (rU/rS) × (CS/CU) × 100
| rU | = | = peak area of the corresponding isomer from the Sample solution |
| rS | = | = peak area of the corresponding isomer from the Standard solution |
| CS | = | = concentration of the appropriate USP Reference Standard in the Standard solution (µg/mL) |
| CU | = | = concentration of the Sample solution (µg/mL) |
[Note—Calculate the percentage of C11H16O2 in the portion of Butylated Hydroxyanisole taken by adding the quantities of the two isomers. ]
Acceptance criteria:
NLT 98.5%
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.01%, determined on a 10-g specimen
:
NMT 0.01%, determined on a 10-g specimen
• Heavy Metals, Method II  231
231 :
NMT 10 ppm
:
NMT 10 ppm
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in well-closed containers.
• USP Reference Standards  11
11
USP 2-tert-Butyl-4-hydroxyanisole RS
C11H16O2 180.25
C11H16O2 180.25
USP 3-tert-Butyl-4-hydroxyanisole RS
C11H16O2 180.25
C11H16O2 180.25
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1717
Pharmacopeial Forum: Volume No. 35(6) Page 1503