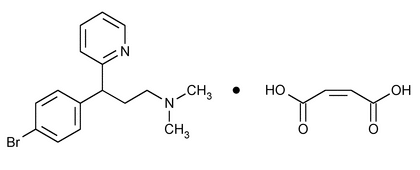
Brompheniramine Maleate
(brome'' fen ir' a meen mal' ee ate).
2-Pyridinepropanamine,
(±)-2-p-Bromo-
» Brompheniramine Maleate, dried at 105 for 3 hours, contains not less than 98.0 percent and not more than 100.5 percent of C16H19BrN2·C4H4O4.
for 3 hours, contains not less than 98.0 percent and not more than 100.5 percent of C16H19BrN2·C4H4O4.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
Solution:
35 µg per mL.
Medium:
methanol.
Melting range  741
741 :
between 130
:
between 130 and 135
and 135 .
.
pH  791
791 :
between 4.0 and 5.0, in a solution (1 in 100).
:
between 4.0 and 5.0, in a solution (1 in 100).
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 3 hours: it loses not more than 0.5% of its weight.
for 3 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.2%.
:
not more than 0.2%.
Related compounds—
Test preparation—
Dissolve about 200 mg of Brompheniramine Maleate in 5 mL of methylene chloride, and mix.
Chromatographic system
(see Chromatography  621
621 )—The gas chromatograph is equipped with a flame-ionization detector and a 1.2-m × 4-mm glass column containing 3% phase G3 on support S1AB. The column temperature is maintained at about 190
)—The gas chromatograph is equipped with a flame-ionization detector and a 1.2-m × 4-mm glass column containing 3% phase G3 on support S1AB. The column temperature is maintained at about 190 , and the injection port and detector temperatures are both maintained at about 250
, and the injection port and detector temperatures are both maintained at about 250 . The carrier gas is dry helium, flowing at a rate adjusted to obtain a retention time of 6 to 7 minutes for the main peak. Chromatograph the Test preparation, record the chromatogram, and determine the peak area as directed for Procedure: the tailing factor for the brompheniramine maleate peak is not more than 1.8.
. The carrier gas is dry helium, flowing at a rate adjusted to obtain a retention time of 6 to 7 minutes for the main peak. Chromatograph the Test preparation, record the chromatogram, and determine the peak area as directed for Procedure: the tailing factor for the brompheniramine maleate peak is not more than 1.8.
Procedure—
Inject a volume (about 1 µL) of the Test preparation into the chromatograph. Record the chromatogram for a total time of not less than twice the retention time of the brompheniramine peak, and measure the areas of the peaks. The total relative area of all extraneous peaks (except that of the solvent peak and maleic acid, if observed) does not exceed 2.0%.
Assay—
Dissolve about 425 mg of Brompheniramine Maleate, previously dried and accurately weighed, in 50 mL of glacial acetic acid. Add 1 drop of crystal violet TS, and titrate with 0.1 N perchloric acid VS to a green endpoint. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 21.77 mg of C16H19BrN2·C4H4O4.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Domenick Vicchio, Ph.D.
Senior Scientific Liaison 1-301-998-6828 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2392
Pharmacopeial Forum: Volume No. 29(5) Page 1431