INTRODUCTION
This general chapter is harmonized with the corresponding texts of the European Pharmacopoeia and/or the Japanese Pharmacopoeia. These pharmacopeias have undertaken not to make any unilateral change to this harmonized chapter. Portions of the present general chapter text that are national USP text, and therefore not part of the harmonized text, are marked with symbols (
 ) to specify this fact.
) to specify this fact.
Pharmaceutical solids as raw materials or as constituents of dosage forms most often come in contact with water during processing and storage. This may occur (a) during crystallization, lyophilization, wet granulation, or spray drying; and (b) because of exposure upon handling and storage to an atmosphere containing water vapor or exposure to other materials in a dosage form that contain water capable of distributing it to other ingredients. Some properties known to be altered by the association of solids with water include rates of chemical degradation in the “solid-state”, crystal growth and dissolution, dispersibility and wetting, powder flow, lubricity, powder compactibility, compact hardness, and microbial contamination.
Although precautions can be taken when water is perceived to be a problem, i.e., eliminating all moisture, reducing contact with the atmosphere, or controlling the relative humidity of the atmosphere, such precautions generally add expense to the process with no guarantee that during the life of the product further problems associated with moisture will be avoided. It is also important to recognize that there are many situations where a certain level of water in a solid is required for proper performance, e.g., powder compaction. It is essential for both reasons, therefore, that as much as possible is known about the effects of moisture on solids before strategies are developed for their handling, storage, and use.
Some of the more critical pieces of required information concerning water–solid interactions are:
— total amount of water present;
— the extent to which adsorption and absorption occur;
— whether or not hydrates form;
— specific surface area of the solid, as well as such properties as degree of crystallinity, degree of porosity, and glass transition and melting temperature;
— site of water interaction, the extent of binding, and the degree of molecular mobility;
— effects of temperature and relative humidity;
— essentially irreversible hydration;
— kinetics of moisture uptake;
— various factors that might influence the rate at which water vapor can be taken up by a solid;
— for water-soluble solids capable of being dissolved by the sorbed water, under which conditions dissolution will take place.
PHYSICAL STATES OF SORBED WATER
Water can physically interact with solids in different ways. It can interact at the surface (adsorption) or it can penetrate the bulk solid structure (absorption). When both adsorption and absorption occur, the term sorption is often used. Adsorption is particularly critical in affecting the properties of solids when the specific surface area is large. Large values of specific surface area are seen with solids having very small particles, as well as with solids having a high degree of intraparticle porosity. Absorption is characterized by an association of water per gram of solid that is much greater than that which can form a monomolecular layer on the available surface, and an amount that is generally independent of the specific surface area.
Most crystalline solids will not absorb water into their bulk structures because of the close packing and high degree of order of the crystal lattice. Indeed, it has been shown that the degree of absorption into solids exhibiting partial crystallinity and partial amorphous structure is often inversely proportional to the degree of crystallinity. With some crystalline solids, however, crystal hydrates may form. These hydrates may exhibit a stoichiometric relationship, in terms of water molecules bound per solid molecule, or they may be non-stoichiometric. Upon dehydration, crystal hydrates may either retain their original crystal structure, lose their crystallinity and become amorphous, or transform into a new anhydrous or less-hydrated crystal form.
Amorphous or partially amorphous solids are capable of taking up significant amounts of water because there is sufficient molecular disorder in the solid to permit penetration, swelling, or dissolution. Such behavior is observed with most amorphous polymers and with small-molecular-mass solids rendered amorphous during preparation, e.g., by lyophilization, or after milling. The introduction of defects into highly crystalline solids will also produce this behavior. The greater the chemical affinity of water for the solid, the greater the total amount that can be absorbed. When water is absorbed by amorphous solids, the bulk properties of the solid can be significantly altered. It is well established, for example, that amorphous solids, depending on the temperature, can exist in at least one of two states: “glassy” or “fluid”; the temperature at which one state transforms into the other is the glass transition temperature, TG.
Water absorbed into the bulk solid structure, by virtue of its effect on the free volume of the solid, can act as an efficient plasticizer and reduce the value of TG. Because the rheological properties of “fluid” and “glassy” states are quite different, i.e., the “fluid” state exhibits much less viscosity as the temperature rises above the glass transition point, it is not surprising that a number of important bulk properties dependent on the rheology of the solid are affected by moisture content. Because amorphous solids are metastable relative to the crystalline form of the material, with small-molecular-mass materials, it is possible for absorbed moisture to initiate reversion of the solid to the crystalline form, particularly if the solid is transformed by the sorbed water to a “fluid” state. This is the basis of “cake collapse” often observed during the lyophilization process. An additional phenomenon noted specifically with water-soluble solids is their tendency to deliquesce, i.e., to dissolve in their own sorbed water, at relative humidities, RHI, in excess of the relative humidity of a saturated solution of the solid, RH0. Deliquescence arises because of the high water solubility of the solid and the significant effect it has on the colligative properties of water. It is a dynamic process that continues to occur as long as RHI is greater than RH0.
The key to understanding the effects water can have on the properties of solids, and vice versa, rests with an understanding of the location of the water molecule and its physical state. More specifically, water associated with solids can exist in a state that is directly bound to the solid, as well as in a state of mobility approaching that of bulk water. This difference in mobility has been observed through such measurements as heat of sorption, freezing point, nuclear magnetic resonance, dielectric properties, and diffusion.
Such changes in mobility have been interpreted as arising because of changes in the thermodynamic state of water as more and more water is sorbed. Thus, water bound directly to a solid is often thought as unavailable to affect the properties of the solid, whereas larger amounts of sorbed water may become more clustered and form water more like that exhibiting solvent properties. In the case of crystal hydrates, the combination of intermolecular forces (hydrogen bonding) and crystal packing can produce very strong water–solid interactions. Recognizing that the presence of water in an amorphous solid can affect the glass transition temperature and hence the physical state of the solid, at low levels of water, most polar amorphous solids are in a highly viscous glassy state because of their high values of TG. Hence, water is “frozen” into the solid structure and is rendered immobile by the high viscosity, e.g., 1013 Pa·s. As the amount of water sorbed increases and TG decreases, approaching ambient temperatures, the glassy state approaches that of a “fluid” state and water mobility along with the mobility of the solid itself increases significantly. At high RH, the degree of water plasticization of the solid can be sufficiently high so that water and the solid can now achieve significant amounts of mobility. In general, therefore, this picture of the nature of sorbed water helps to explain the rather significant effect moisture can have on a number of bulk properties of solids such as chemical reactivity and mechanical deformation. It suggests strongly that methods of evaluating chemical and physical stability of solids and solid dosage forms take into account the effects water can have on the solid when it is sorbed, particularly when it enters the solid structure and acts as a plasticizer.
Rates of Water Uptake
The rate and extent to which solids exposed to the atmosphere might either sorb or desorb water vapor can be a critical factor in the handling of solids. Even the simple act of weighing out samples of solid on an analytical balance and the exposure, therefore, of a thin layer of powder to the atmosphere for a few minutes can lead to significant error in, for example, the estimation of loss on drying values. It is well established that water-soluble solids exposed to relative humidities above that exhibited by a saturated solution of that solid will spontaneously dissolve via deliquescence and continue to dissolve over a long time period. The rate of water uptake in general depends on a number of parameters not found to be critical in equilibrium measurements because rates of sorption are primarily mass-transfer controlled with some contributions from heat-transfer mechanisms. Thus, factors such as vapor diffusion coefficients in air and in the solid, convective airflow, and the surface area and geometry of the solid bed and surrounding environment, can play an important role. Indeed, the method used to make measurements can often be the rate-determining factor because of these environmental and geometric factors.
DETERMINATION OF SORPTION–DESORPTION ISOTHERMS
Principle
The tendency to take up water vapor is best assessed by measuring sorption or desorption as a function of relative humidity, at constant temperature, and under conditions where sorption or desorption is essentially occurring independently of time, i.e., equilibrium. Relative humidity, RH, is defined by the following equation:
RH = (PC × 100)/P0
PC = pressure of water vapor in the system
P0 = saturation pressure of water vapor under the same conditions
The ratio PC/P0 is referred to as the relative pressure. Sorption or water uptake is best assessed starting with dried samples and subjecting them to a known relative humidity. Desorption is studied by beginning with a system already containing sorbed water and reducing the relative humidity. As the name indicates, the sorption–desorption isotherm is valid only for the reference temperature, hence a special isotherm exists for each temperature. Ordinarily, at equilibrium, moisture content at a particular relative humidity must be the same, whether determined from sorption or desorption measurements. However, it is common to see sorption–desorption hysteresis.
Methods
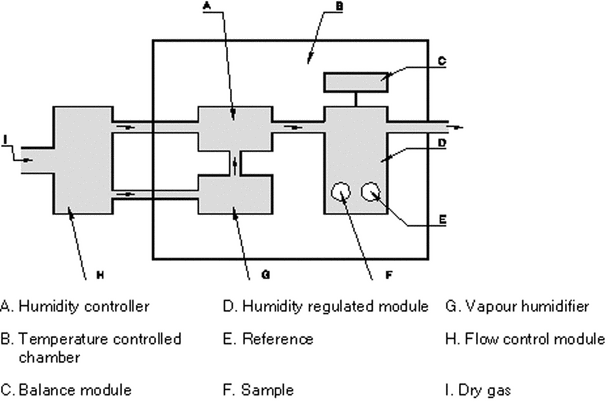
Samples may be stored in chambers at various relative humidities. The mass gained or lost for each sample is then measured. The major advantage of this method is convenience, while the major disadvantages are the slow rate of reaching constant mass, particularly at high relative humidities, and the error introduced in opening and closing the chamber for weighing. Dynamic gravimetric water sorption systems allow the on-line weighing of a sample in a controlled system to assess the interaction of the material with moisture at various programmable levels of relative humidity at a constant temperature. The major benefit of a controlled system is that isothermal conditions can be more reliably established and that the dynamic response of the sample to changing conditions can be monitored (see Figure 1).
Figure 1. Example of an apparatus for the determination of the water sorption (other designs are possible).
Data points for the determination of the sorption isotherm (e.g., from 0% to approximately 95% RH, noncondensing) are only taken after a sufficiently constant signal indicates that the sample has reached equilibrium at a given level of humidity. In some cases (e.g., deliquescence), the maximum time may be restricted although the equilibrium level is not reached. The apparatus must adequately control the temperature to ensure a good baseline stability as well as accurate control of the relative humidity generation. The required relative humidities can be generated, e.g., by accurately mixing dry and saturated vapor gas with flow controllers. The electrostatic behavior of the powder must also be considered. The verification of the temperature and the relative humidity (controlled with, for example, a certified hygrometer, certified salt solutions, or deliquescence points of certified salts over an adequate range) must be consistent with the instrument specification. The balance must provide a sufficient mass resolution and long-term stability.
It is also possible to measure amounts of water uptake not detectable gravimetrically using volumetric techniques. In the case of adsorption, to improve sensitivity, the specific surface area of the sample can be increased by reducing particle size or by using larger samples to increase the total area. It is important, however, that such comminution of the solid does not alter the surface structure of the solid or render it more amorphous or otherwise less ordered in crystallinity. For absorption, where water uptake is independent of specific surface area, only increasing sample size will help. Increasing sample size, however, will increase the time to establish some type of equilibrium. To establish accurate values, it is important to get desolvation of the sample as thoroughly as possible. Higher temperatures and lower pressures (vacuum) facilitate this process; however, care must be taken to note any adverse effects this might have on the solid such as dehydration, chemical degradation, or sublimation. Using higher temperatures to induce desorption, as in a thermogravimetric apparatus, likewise must be carefully carried out because of these possible pitfalls.
Report and Interpretation of the Data
Sorption data are usually reported as a graph of the apparent mass change in percent of the mass of the dry sample as a function of relative humidity or time. Sorption isotherms are reported both in tabular form and as a graph. The measurement method must be traceable with the data.
Adsorption–desorption hysteresis can be interpreted, for example, in terms of the porosity of the sample, its state of agglomeration (capillary condensation), the formation of hydrates, polymorphic change, or liquefying of the sample. Certain types of systems, particularly those with microporous solids and amorphous solids, are capable of sorbing large amounts of water vapor. Here, the amount of water associated with the solid as relative humidity is decreased, is greater than the amount that originally sorbed as the relative humidity was increased. For microporous solids, vapor adsorption–desorption hysteresis is an equilibrium phenomenon associated with the process of capillary condensation. This takes place because of the high degree of irregular curvature of the micropores and the fact that they “fill” (adsorption) and “empty” (desorption) under different equilibrium conditions. For nonporous solids capable of absorbing water, hysteresis occurs because of a change in the degree of vapor–solid interaction due to a change in the equilibrium state of the solid, e.g., conformation of polymer chains, or because the time scale for structural equilibrium is longer than the time scale for water desorption. In measuring sorption–desorption isotherms, it is therefore important to establish that something close to an equilibrium state has been reached. Particularly with hydrophilic polymers at high relative humidities, the establishment of water sorption or desorption values independent of time is quite difficult, because one is usually dealing with a polymer plasticized into its “fluid” state, where the solid is undergoing significant change.
In the case of crystal hydrate formation, the plot of water uptake versus pressure or relative humidity will in these cases exhibit a sharp increase in uptake at a particular pressure and the amount of water taken up will usually exhibit a stoichiometric mole:mole ratio of water to solid. In some cases, however, crystal hydrates will not appear to undergo a phase change or the anhydrous form will appear amorphous. Consequently, water sorption or desorption may appear more like that seen with adsorption processes. X-ray crystallographic analysis and thermal analysis are particularly useful for the study of such systems.
For situations where water vapor adsorption occurs predominantly, it is very helpful to measure the specific surface area of the solid by an independent method and to express adsorption as mass of water sorbed per unit area of solid surface. This can be very useful in assessing the possible importance of water sorption in affecting solid properties. For example, 0.5% m/m uptake of water could hardly cover the bare surface of 100 m2/g, while for 1.0 m2/g this amounts to 100 times more surface coverage. In the case of pharmaceutical solids that have a specific surface area in the range of 0.01 m2/g to 10 m2/g, what appears to be low water content could represent a significant amount of water for the available surface. Because the “dry surface area” is not a factor in absorption, sorption of water with amorphous or partially amorphous solids can be expressed on the basis of unit mass corrected for crystallinity, when the crystal form does not sorb significant amounts of water relative to the amorphous regions.
DETERMINATION OF THE WATER ACTIVITY
Principle
Water activity, AW, is the ratio of vapor pressure of water in the product (P) to saturation pressure of water vapor (P0) at the same temperature. It is numerically equal to 1/100 of the relative humidity (RH) generated by the product in a closed system. RH can be calculated from direct measurements of partial vapor pressure or dew point, or from indirect measurement by sensors whose physical or electric characteristics are altered by the RH to which they are exposed. Ignoring activity coefficients, the relationship between AW and equilibrium relative humidity (ERH) are represented by the following equations:
AW = P/P0
ERH(%) = AW × 100
Method
The water activity is determined by placing the sample in a small airtight cup inside which the equilibrium between the water in the solid and the headspace can be established. The volume of the headspace must be small in relation to the sample volume in order not to change the sorption state of the sample during the test. The equilibration as a thermodynamic process takes time but may be accelerated by forced circulation within the cell. The acquired water activity value is only valid for the simultaneously determined temperature. This requires a precise temperature-measuring device as part of the equipment. Furthermore, the probe must be thermally insulated to guarantee a constant temperature during the test. The sensor measuring the humidity of the headspace air above the sample is a key component. Theoretically, all types of hygrometers can be used, but for analytical purposes miniaturization and robustness are a precondition. The AW measurement may be conducted using the dew point/chilled mirror method.1 A polished, chilled mirror is used as a condensing surface. The cooling system is electronically linked to a photoelectric cell into which light is reflected from the condensing mirror. An air stream, in equilibrium with the test sample, is directed at the mirror which cools until condensation occurs on the mirror. The temperature at which this condensation begins is the dew point from which the ERH is determined. Commercially available instruments using the dew point/chilled mirror method or other technologies need to be evaluated for suitability, validated, and calibrated when used to make water activity determinations.
These instruments are typically calibrated over an adequate range, for example, using some saturated salt solutions at 25 such as those listed in Table 1.
such as those listed in Table 1.
Table 1. Standard Saturated Salt Solutions
| Saturated Salt Solutions at 25 |
ERH (%) |
AW |
|---|---|---|
| Potassium sulfate (K2SO4) | 97.3 | 0.973 |
| Barium chloride (BaCl2) | 90.2 | 0.902 |
| Sodium chloride (NaCl) | 75.3 | 0.753 |
| Magnesium nitrate (Mg(NO3)2) | 52.9 | 0.529 |
| Magnesium chloride (MgCl2) | 32.8 | 0.328 |
| Lithium chloride (LiCl2) | 11.2 | 0.112 |
1
AOAC International Official Method 978.18.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| General Chapter | Antonio Hernandez-Cardoso, M.Sc.
Senior Scientific Liaison 1-301-816-8308 |
(GCPA2010) General Chapters - Physical Analysis |
USP35–NF30 Page 934
Pharmacopeial Forum: Volume No. 36(4) Page 1063