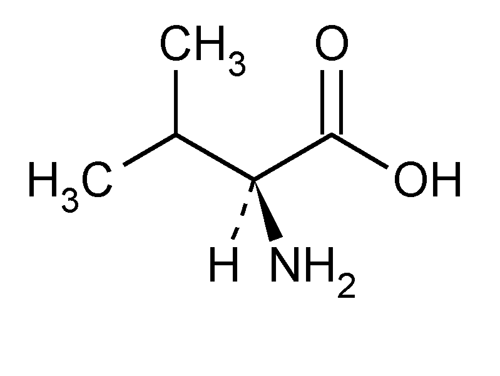
Valine
» Valine contains not less than 98.5 percent and not more than 101.5 percent of C5H11NO2, as l-valine, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
Identification,
Infrared Absorption  197K
197K .
.
Specific rotation  781S
781S :
between +26.6
:
between +26.6 and +28.8
and +28.8 .
.
Test solution:
80 mg per mL, in 6 N hydrochloric acid.
pH  791
791 :
between 5.5 and 7.0, in a solution (1 in 20).
:
between 5.5 and 7.0, in a solution (1 in 20).
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 3 hours: it loses not more than 0.3% of its weight.
for 3 hours: it loses not more than 0.3% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Chloride  221
221 —
A 0.73-g portion shows no more chloride than corresponds to 0.50 mL of 0.020 N hydrochloric acid (0.05%).
—
A 0.73-g portion shows no more chloride than corresponds to 0.50 mL of 0.020 N hydrochloric acid (0.05%).
Sulfate  221
221 —
A 0.33-g portion shows no more sulfate than corresponds to 0.10 mL of 0.020 N sulfuric acid (0.03%).
—
A 0.33-g portion shows no more sulfate than corresponds to 0.10 mL of 0.020 N sulfuric acid (0.03%).
Iron  241
241 :
0.003%.
:
0.003%.
Heavy metals, Method I  231
231 :
0.0015%.
:
0.0015%.
Chromatographic purity—
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture.
Test solution—
Dissolve an accurately weighed quantity of Valine in 2 N hydrochloric acid to obtain a solution having a concentration of 10 mg per mL. Apply 5 µL.
Standard solution—
Dissolve an accurately weighed quantity of USP l-Valine RS in 0.1 N hydrochloric acid to obtain a solution having a known concentration of about 0.05 mg per mL. Apply 5 µL. [note—This solution has a concentration equivalent to about 0.5% of that of the Test solution.]
System suitability solution—
Prepare a solution in 0.1 N hydrochloric acid containing 0.4 mg each of USP l-Valine RS and USP l-Phenylalanine RS per mL. Apply 5 µL.
Spray reagent—
Dissolve 0.2 g of ninhydrin in 100 mL of a mixture of butyl alcohol and 2 N acetic acid (95:5).
Developing solvent system—
Prepare a mixture of butyl alcohol, glacial acetic acid, and water (60:20:20).
Procedure—
Proceed as directed for Thin-Layer Chromatography under Chromatography  621
621 . After air-drying the plate, spray with Spray reagent, and heat between 100
. After air-drying the plate, spray with Spray reagent, and heat between 100 and 105
and 105 for about 15 minutes. Examine the plate under white light. The chromatogram obtained from the System suitability solution exhibits two clearly separated spots. Any secondary spot in the chromatogram obtained from the Test solution is not larger or more intense than the principal spot in the chromatogram obtained from the Standard solution: not more than 0.5% of any individual impurity is found; and not more than 2.0% of total impurities is found.
for about 15 minutes. Examine the plate under white light. The chromatogram obtained from the System suitability solution exhibits two clearly separated spots. Any secondary spot in the chromatogram obtained from the Test solution is not larger or more intense than the principal spot in the chromatogram obtained from the Standard solution: not more than 0.5% of any individual impurity is found; and not more than 2.0% of total impurities is found.
Assay—
Transfer about 110 mg of Valine, accurately weighed, to a 125-mL flask, dissolve in a mixture of 3 mL of formic acid and 50 mL of glacial acetic acid, and titrate with 0.1 N perchloric acid VS, determining the endpoint potentiometrically. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 11.72 mg of C5H11NO2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Curtis Phinney
1-301-816-8540 |
(DSN05) Dietary Supplements - Non-Botanicals |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3837
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.