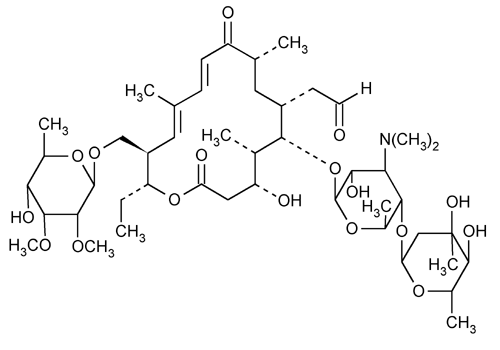
Tylosin
C46H77NO17




 916.1
916.1
(10E,12E)-(3R,4S,5S,6R,8R,14S,15R)-14-[(6-deoxy-2,3-di-O-methyl- -d-allopyranosyl)oxymethyl]-5-[[3,6-dideoxy-4-O-(2,6-dideoxy-3-C-methyl-
-d-allopyranosyl)oxymethyl]-5-[[3,6-dideoxy-4-O-(2,6-dideoxy-3-C-methyl- -l-ribo-hexopyranosyl)-3-dimethylamino-
-l-ribo-hexopyranosyl)-3-dimethylamino- -d-glucopyranosyl]oxy]-6-formylmethyl-3-hydroxy-4,8,12-trimethyl-9-oxoheptadeca-10,12-dien-15-olide.
-d-glucopyranosyl]oxy]-6-formylmethyl-3-hydroxy-4,8,12-trimethyl-9-oxoheptadeca-10,12-dien-15-olide.
Tylosin A

 [1401-69-0].
[1401-69-0].
(10E,12E)-(3R,4S,5S,6R,8R,14S,15R)-14-[(6-deoxy-2,3-di-O-methyl-
Tylosin A
» Tylosin is the macrolide antibiotic substance, or the mixture of such substances, produced by the growth of Streptomyces fradiae, or by any other means. Its potency is not less than 900 µg of tylosin per mg, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers, protected from light, moisture, and excessive heat.
Labeling—
Label it to indicate that it is for use in animals only.
USP Reference standards  11
11 —
—
USP Tylosin RS.
USP Tylosin RS.
Identification—
A:
Ultraviolet Absorption  197U
197U .
.
Acid solution—
Transfer about 50 mg of Tylosin, accurately weighed, to a 100-mL volumetric flask, add 10 mL of 2 N hydrochloric acid, dilute with water to volume, and mix. Transfer 5.0 mL of this solution to a second 100-mL volumetric flask, dilute with water to volume, and mix. Absorptivity at 290 nm is 22.5 ± 2.5, calculated on the dried basis.
Alkaline solution—
To 10.0 mL of the final Acid solution add 1.0 mL of 2 N sodium hydroxide, and heat on a water bath for 20 minutes. Cool to room temperature: an absorption maximum is observed at about 332 nm.
B:
The retention time of the major peak for tylosin A in the chromatogram of the Test solution corresponds to that in the chromatogram of the Standard solution, as obtained in the test for Content of tylosins.
Loss on drying  731
731 —
Dry about 1 g, accurately weighed, in vacuum at a pressure of not more than 5 mm of mercury at 60
—
Dry about 1 g, accurately weighed, in vacuum at a pressure of not more than 5 mm of mercury at 60 for 3 hours: it loses not more than 5% of its weight.
for 3 hours: it loses not more than 5% of its weight.
Residue on ignition  281
281 :
not more than 3.0%, the charred residue being moistened with 2 mL of nitric acid and 5 drops of sulfuric acid.
:
not more than 3.0%, the charred residue being moistened with 2 mL of nitric acid and 5 drops of sulfuric acid.
Heavy metals, Method II  231
231 :
0.003%.
:
0.003%.
Limit of tyramine—
Transfer 100 mg of it to a 25-mL volumetric flask, add 5.0 mL of 0.03 M phosphoric acid, and swirl to dissolve (Test solution). Transfer 5.0 mL of a solution containing 70 µg of tyramine per mL in 0.03 M phosphoric acid to a 25-mL volumetric flask (Standard solution). Transfer 5 mL of 0.03 M phosphoric acid to a 25-mL volumetric flask to provide the blank. Concurrently add to each flask 1.0 mL of a mixture of pyridine and 2.0 mL of filtered ninhydrin solution (1 in 25). Cover the flasks lightly with glass or aluminum foil caps, and heat in a water bath at 85 for not less than 20 minutes. Cool rapidly to room temperature, dilute with water to volume, and mix. Promptly determine the absorbances of the solutions from the Test solution and the Standard solution at the wavelength of maximum absorbance at about 570 nm, using the solution from the blank to zero the instrument. The absorbance of the solution from the Test solution is not greater than that of the solution from the Standard solution (0.35% of tyramine). In a valid test the solution from the Standard solution exhibits a dark blue color.
for not less than 20 minutes. Cool rapidly to room temperature, dilute with water to volume, and mix. Promptly determine the absorbances of the solutions from the Test solution and the Standard solution at the wavelength of maximum absorbance at about 570 nm, using the solution from the blank to zero the instrument. The absorbance of the solution from the Test solution is not greater than that of the solution from the Standard solution (0.35% of tyramine). In a valid test the solution from the Standard solution exhibits a dark blue color.
Content of tylosins—
Mobile phase—
Prepare a mixture of filtered 2 M sodium perchlorate, previously adjusted with 1 N hydrochloric acid to a pH of 2.5 ± 0.1, and acetonitrile (60:40). Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Standard solution—
Transfer about 30 mg of USP Tylosin RS, accurately weighed, to a 100-mL volumetric flask, add 10 mL of methanol, and swirl to dissolve. Dilute with water to volume, and mix.
Test solution—
Transfer about 30 mg of Tylosin, accurately weighed, to a 100-mL volumetric flask, add 10 mL of methanol, and swirl to dissolve. Dilute with water to volume, and mix.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 280-nm detector, a 4.6-mm × 20-cm column that contains 5-µm packing L1. The flow rate is about 0.7 mL per minute. Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the resolution, R, between the tylosin D peak and the tylosin A peak is not less than 2, the tailing factor is not more than 1.5, and the relative standard deviation for replicate injections is not more than 2%.
)—
The liquid chromatograph is equipped with a 280-nm detector, a 4.6-mm × 20-cm column that contains 5-µm packing L1. The flow rate is about 0.7 mL per minute. Chromatograph the Standard solution, and record the peak responses as directed for Procedure: the resolution, R, between the tylosin D peak and the tylosin A peak is not less than 2, the tailing factor is not more than 1.5, and the relative standard deviation for replicate injections is not more than 2%.
Procedure—
Separately inject equal volumes (about 20 µL) of the Standard solution and the Test solution into the chromatograph, record the chromatograms over a period of time 1.5 times the elution time of the main tylosin A peak, and measure the peak areas for all the peaks. The relative retention times are about 0.5 for tylosin C, 0.7 for tylosin B, 0.9 for tylosin D, and 1.0 for tylosin A. Calculate the percentages of tylosin A, tylosin B, tylosin C, and tylosin D in the Tylosin taken by the formula:
100(ri / rs)
in which ri is the area of the tylosin A peak, the tylosin B peak, tylosin C peak, or the tylosin D peak, as appropriate, in the chromatogram obtained from the Test solution, and rs is the sum of the areas of all of the peaks in the chromatogram obtained from the Test solution: the content of tylosin A is not less than 80% and the sum of the contents of tylosin A, tylosin B, tylosin C, and tylosin D is not less than 95%.
Assay—
Proceed as directed for Tylosin under Antibiotics—Microbial Assays  81
81 . Prepare the Test Dilution as follows. Transfer about 250 mg of Tylosin, accurately weighed, to a 500-mL volumetric flask, add 50 mL of methanol, and swirl to dissolve. Dilute with Buffer No. 3 to volume, and mix. Transfer 4.0 mL of this solution to a second 500-mL volumetric flask, dilute with a mixture of Buffer No. 3 and methanol (1:1), and mix. This solution contains about 4 µg of tylosin per mL.
. Prepare the Test Dilution as follows. Transfer about 250 mg of Tylosin, accurately weighed, to a 500-mL volumetric flask, add 50 mL of methanol, and swirl to dissolve. Dilute with Buffer No. 3 to volume, and mix. Transfer 4.0 mL of this solution to a second 500-mL volumetric flask, dilute with a mixture of Buffer No. 3 and methanol (1:1), and mix. This solution contains about 4 µg of tylosin per mL.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Ian DeVeau, Ph.D.
Director, Veterinary Drugs and Radiopharmaceuticals 1-301-816-8178 |
(VET05) Veterinary Drugs 05 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3826
Pharmacopeial Forum: Volume No. 27(4) Page 2780
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.