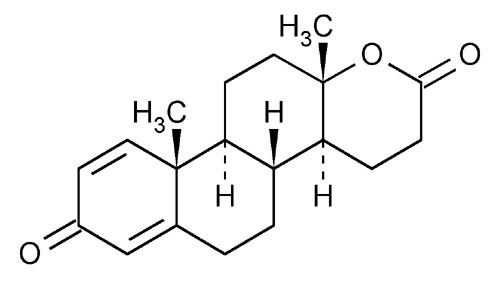
Testolactone
d-Homo-17a-oxaandrosta-1,4-diene-3,17-dione.
13-Hydroxy-3-oxo-13,17-secoandrosta-1,4-dien-17-oic acid
» Testolactone contains not less than 95.0 percent and not more than 105.0 percent of C19H24O3, calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
USP Reference standards  11
11 —
—
USP Testolactone RS.
USP Testolactone RS.
Identification—
Solution:
10 µg per mL.
Medium:
methanol.
Loss on drying  731
731 —
Dry it in vacuum at 100
—
Dry it in vacuum at 100 for 3 hours: it loses not more than 1.0% of its weight.
for 3 hours: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method II  231
231 :
0.003%.
:
0.003%.
Chromatographic purity—
Standard preparation—
Transfer 10 mg of USP Testolactone RS, accurately weighed, to a 50-mL volumetric flask, dissolve in acetone, dilute with acetone to volume, and mix.
Test preparation—
Transfer 250 mg of Testolactone, accurately weighed, to a 50-mL volumetric flask, dissolve in acetone, dilute with acetone to volume, and mix.
Procedure—
Coat a 20- × 20-cm thin-layer chromatographic plate (see Chromatography  621
621 ) with a 0.25-mm layer of chromatographic silica gel mixture, dry for 15 minutes at room temperature, heat at 105
) with a 0.25-mm layer of chromatographic silica gel mixture, dry for 15 minutes at room temperature, heat at 105 for 1 hour, and cool in a desiccator. Divide the area of the plate into three approximately equal sections, the left and right sections to be used for the Test preparation and the Standard preparation, respectively, and the center section for the blank. Apply 10 µL of the Standard preparation and 20 µL of the Test preparation 2.5 cm from the bottom of the designated sections of the plate, and dry the spots with a current of air. Develop the chromatogram in a solvent system consisting of a mixture of butyl acetate and acetone (4:1) until the solvent front has moved to within about 1 cm of the top of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow the solvent to evaporate. Locate the spots on the plate by viewing under short-wavelength UV light: the RF value of the principal spot obtained from the Test preparation corresponds to that obtained from the Standard preparation, not more than two impurities are found in the chromatogram of the Test preparation, and the size and color of the spot representing any impurity obtained from the Test preparation are not greater or more intense than those of the principal spot obtained from the Standard preparation.
for 1 hour, and cool in a desiccator. Divide the area of the plate into three approximately equal sections, the left and right sections to be used for the Test preparation and the Standard preparation, respectively, and the center section for the blank. Apply 10 µL of the Standard preparation and 20 µL of the Test preparation 2.5 cm from the bottom of the designated sections of the plate, and dry the spots with a current of air. Develop the chromatogram in a solvent system consisting of a mixture of butyl acetate and acetone (4:1) until the solvent front has moved to within about 1 cm of the top of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow the solvent to evaporate. Locate the spots on the plate by viewing under short-wavelength UV light: the RF value of the principal spot obtained from the Test preparation corresponds to that obtained from the Standard preparation, not more than two impurities are found in the chromatogram of the Test preparation, and the size and color of the spot representing any impurity obtained from the Test preparation are not greater or more intense than those of the principal spot obtained from the Standard preparation.
Ordinary impurities  466
466 —
—
Test solution:
methanol.
Standard solution:
methanol.
Eluant:
a mixture of butyl acetate and acetone (4:1).
Visualization:
6.
Assay—
Isoniazid reagent—
Dissolve 1.0 g of isoniazid in about 500 mL of methanol, add 1.25 mL of hydrochloric acid, dilute with methanol to 1000 mL, and mix.
Standard preparation—
Dissolve a suitable quantity of USP Testolactone RS, accurately weighed, in chloroform, and prepare, by quantitative, and stepwise dilution if necessary, a solution in chloroform having a known concentration of about 30 µg per mL.
Assay preparation—
Transfer about 60 mg of Testolactone, accurately weighed, to a 100-mL volumetric flask, dissolve in chloroform, dilute with chloroform to volume, and mix. Transfer 5.0 mL of this solution to a second 100-mL volumetric flask, dilute with chloroform to volume, and mix.
Procedure—
Transfer 5.0 mL each of the Standard preparation, the Assay preparation, and chloroform to provide the blank, to separate 25-mL volumetric flasks, add 10.0 mL of Isoniazid reagent to each flask, and mix. Place the flasks in a water bath maintained at a temperature of 55 ± 2 , and allow them to stand for 70 minutes. Cool, dilute each solution with chloroform to volume, and mix. Determine the absorbances of the solutions in 1-cm cells at the wavelength of maximum absorbance at about 415 nm, with a suitable spectrophotometer, against the blank. Calculate the quantity, in mg, of C19H24O3 in the Testolactone taken by the formula:
, and allow them to stand for 70 minutes. Cool, dilute each solution with chloroform to volume, and mix. Determine the absorbances of the solutions in 1-cm cells at the wavelength of maximum absorbance at about 415 nm, with a suitable spectrophotometer, against the blank. Calculate the quantity, in mg, of C19H24O3 in the Testolactone taken by the formula:
2C(AU / AS)
in which C is the concentration, in µg per mL, of USP Testolactone RS in the Standard preparation, and AU and AS are the absorbances of the solutions from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Feiwen Mao, M.S.
Scientist 1-301-816-8320 |
(MDOOD05) Monograph Development-Ophthalmics Oncologics and Dermatologicals |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3683
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.