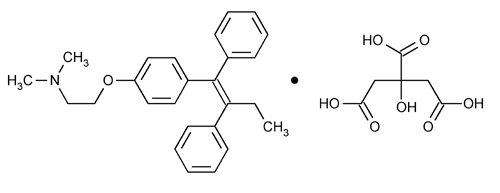
Tamoxifen Citrate
Ethanamine, 2-[4-(1,2-diphenyl-1-butenyl)phenoxy]-N,N-dimethyl, (Z)-, 2-hydroxy-1,2,3-propanetricarboxylate (1:1).
(Z)-2-[p-(1,2-Diphenyl-1-butenyl)phenoxy]-N,N-dimethylethylamine citrate (1:1)
» Tamoxifen Citrate contains not less than 99.0 percent and not more than 101.0 percent of C26H29NO·C6H8O7, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers.
Identification—
Solution:
20 µg per mL.
Medium:
methanol.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 4 hours: it loses not more than 0.5% of its weight.
for 4 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.2%.
:
not more than 0.2%.
Limit of E-isomer—
Mobile phase—
Prepare a methanol solution containing, in each liter, 320 mL of water, 2 mL of glacial acetic acid, and 1.08 g of sodium 1-octanesulfonate.
Standard preparation—
Dissolve a suitable quantity, accurately weighed, of USP Tamoxifen Citrate RS in Mobile phase to obtain a solution having a known concentration of about 600 µg per mL.
Test preparation—
Using about 30 mg of Tamoxifen Citrate, accurately weighed, proceed as directed under Standard preparation.
Chromatographic system
(see Chromatography  621
621 )—The liquid chromatograph is equipped with a 254-nm detector and a 4-mm × 30-cm column that contains packing L11. The flow rate is about 0.7 mL per minute. Chromatograph five replicate injections of the Standard preparation, and record the responses of the major peak: the relative standard deviation is not more than 3.0%, and the relative retention time of the minor E-isomer peak to that of the Z-isomer peak is not greater than 0.93.
)—The liquid chromatograph is equipped with a 254-nm detector and a 4-mm × 30-cm column that contains packing L11. The flow rate is about 0.7 mL per minute. Chromatograph five replicate injections of the Standard preparation, and record the responses of the major peak: the relative standard deviation is not more than 3.0%, and the relative retention time of the minor E-isomer peak to that of the Z-isomer peak is not greater than 0.93.
Procedure—
Separately introduce equal volumes (about 20 µL) of the Test preparation and the Standard preparation into the liquid chromatograph by means of a suitable sampling valve. Measure the minor peak responses for the E-isomer obtained from the Standard preparation and the Assay preparation. Calculate the quantity, in mg, of E-isomer (C26H29NO·C6H8O7) in the portion of Tamoxifen Citrate taken by the formula:
0.05C(rU / rS)
in which C is the concentration, in µg per mL, of the E-isomer as the citrate, based on its declared content in USP Tamoxifen Citrate RS in the Standard preparation, and the rU and rS are the minor peak responses obtained from the Assay preparation and the Standard preparation, respectively. The E-isomer content is not more than 0.3% of tamoxifen citrate (C26H29NO·C6H8O7).
Iron  241
241 —
Accurately weigh 1.0 g, and transfer to a suitable crucible. Add sufficient sulfuric acid to wet the substance, and carefully ignite at a low temperature until thoroughly charred. (The crucible may be loosely covered with a suitable lid during the charring.) Add to the carbonized mass 2 mL of nitric acid and 5 drops of sulfuric acid, and heat cautiously until white fumes no longer are evolved. Ignite, preferably in a muffle furnace, at 500
—
Accurately weigh 1.0 g, and transfer to a suitable crucible. Add sufficient sulfuric acid to wet the substance, and carefully ignite at a low temperature until thoroughly charred. (The crucible may be loosely covered with a suitable lid during the charring.) Add to the carbonized mass 2 mL of nitric acid and 5 drops of sulfuric acid, and heat cautiously until white fumes no longer are evolved. Ignite, preferably in a muffle furnace, at 500 to 600
to 600 , until the carbon is completely burned off. Cool, add 10 mL of warm 0.1 N hydrochloric acid, and digest for about 5 minutes. Transfer the contents of the crucible with the aid of small portions of water to a 50-mL volumetric flask, dilute with water to volume, and mix. Pipet 10 mL from the volumetric flask into a color-comparison tube, dilute with water to 45 mL, add 2 mL of hydrochloric acid, and mix. The limit is 0.005%.
, until the carbon is completely burned off. Cool, add 10 mL of warm 0.1 N hydrochloric acid, and digest for about 5 minutes. Transfer the contents of the crucible with the aid of small portions of water to a 50-mL volumetric flask, dilute with water to volume, and mix. Pipet 10 mL from the volumetric flask into a color-comparison tube, dilute with water to 45 mL, add 2 mL of hydrochloric acid, and mix. The limit is 0.005%.
Heavy metals, Method II  231
231 :
0.001%.
:
0.001%.
Related impurities—
Test preparation A—
Disperse about 3 g in 100 mL of water in a separator. Over a 10-minute period add 50 mL of 0.5 N sodium hydroxide, with mixing. Extract with two 50-mL portions of ether, and combine the extracts. Wash with 20 mL of water, remove the water layer, and dry the ether layer over anhydrous sodium sulfate. Evaporate the ether layer under nitrogen, and dry in vacuum at room temperature for 2 hours. Accurately weigh 1.5 g of the residue into a 10-mL volumetric flask, add 5.0 mL of a mixture of 5 volumes of acetic anhydride and 95 volumes of pyridine, and heat at 60 for 10 to 15 minutes. Cool, dilute with the same solvent mixture to volume, and mix.
for 10 to 15 minutes. Cool, dilute with the same solvent mixture to volume, and mix.
Test preparation B—
Using the same acetic anhydride-pyridine mixture, prepare a 1:200 dilution of Test preparation A.
Chromatographic system
(see Chromatography  621
621 )—Typically, the gas chromatograph is equipped with a flame-ionization detector, and contains a 4-mm × 1-m glass column packed with 5% liquid phase G17 on 100- to 120-mesh support S1AB conditioned at 300
)—Typically, the gas chromatograph is equipped with a flame-ionization detector, and contains a 4-mm × 1-m glass column packed with 5% liquid phase G17 on 100- to 120-mesh support S1AB conditioned at 300 for 24 hours. The column and injection port temperatures are maintained at about 260
for 24 hours. The column and injection port temperatures are maintained at about 260 and the detector temperature at about 300
and the detector temperature at about 300 . Dry helium is used as the carrier gas at a flow rate of about 60 mL per minute. In a suitable chromatogram, five replicate injections of Test preparation B show a relative standard deviation of not more than 3.0%.
. Dry helium is used as the carrier gas at a flow rate of about 60 mL per minute. In a suitable chromatogram, five replicate injections of Test preparation B show a relative standard deviation of not more than 3.0%.
Procedure—
Inject equal portions (about 2 µL), accurately measured, of Test preparation A and Test preparation B into the chromatograph, and record the chromatograms from 0.1 to 5.0, relative to the retention time of the major peak. Measure the individual areas of the peaks other than those produced by the solvent and the tamoxifen on the chromatograms obtained from Test preparation A, and calculate their sum. No single peak area is greater than the total area of the tamoxifen peak on the chromatogram obtained from Test preparation B (0.5%), and the sum of the peak areas is not greater than twice the total area of the tamoxifen peak on the chromatogram obtained from Test preparation B (1.0%).
Assay—
Weigh accurately about 1 g of Tamoxifen Citrate, and dissolve in 150 mL of glacial acetic acid. Titrate the solution with 0.1 N perchloric acid VS, determining the endpoint potentiometrically, using a glass indicator electrode and a silver-silver chloride reference electrode. Each mL of 0.1 N perchloric acid is equivalent to 56.36 mg of C26H29NO·C6H8O7.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Daniel K. Bempong, Ph.D.
Senior Scientist 1-301-816-8143 |
(MDPS05) Monograph Development-Pulmonary and Steroids |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3651
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.