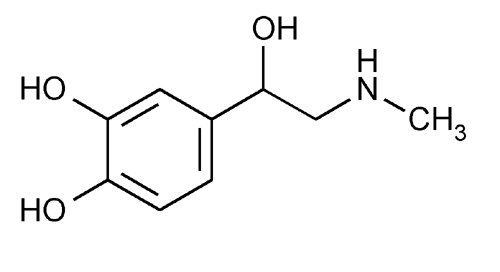
Racepinephrine
1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, (±)-.
(±)-3,4-Dihydroxy-
» Racepinephrine is a racemic mixture of the enantiomorphs of epinephrine. It contains not less than 97.0 percent and not more than 102.0 percent of C9H13NO3, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
To 5 mL of pH 4.0 acid phthalate buffer (see Buffer Solutions in the section Reagents, Indicators, and Solutions) add 0.5 mL of a solution of Racepinephrine (1 in 1000) and 1.0 mL of 0.1 N iodine. Mix, and allow to stand for 5 minutes. Add 2 mL of sodium thiosulfate solution (1 in 40): a deep red color is produced.
Loss on drying  731
731 —
Dry it in vacuum over silica gel for 18 hours: it loses not more than 2.0% of its weight.
—
Dry it in vacuum over silica gel for 18 hours: it loses not more than 2.0% of its weight.
Residue on ignition  281
281 :
not more than 0.5%.
:
not more than 0.5%.
Limit of adrenalone—
Its absorptivity (see Spectrophotometry and Light-scattering  851
851 ) at 310 nm, determined in a solution in dilute hydrochloric acid (1 in 200) containing 2 mg per mL, is not more than 0.2.
) at 310 nm, determined in a solution in dilute hydrochloric acid (1 in 200) containing 2 mg per mL, is not more than 0.2.
Limit of norepinephrine—
Epinephrine standard solution—
Dilute with methanol an accurately measured volume of a solution of USP Epinephrine Bitartrate RS in formic acid containing about 364 mg per mL to obtain a solution having a concentration of about 20 mg per mL.
Norepinephrine standard solution—
Dilute with methanol an accurately measured volume of a solution of USP Norepinephrine Bitartrate RS in formic acid containing 16 mg per mL to obtain a solution having a known concentration of 1.6 mg per mL.
Test solution—
Dissolve 200 mg of Racepinephrine in 1.0 mL of formic acid, dilute with methanol to 10.0 mL, and mix.
Procedure—
Apply 5-µL portions of Epinephrine standard solution, Norepinephrine standard solution, and Test solution to a suitable thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel mixture. Allow to dry, and develop the chromatogram in an unsaturated tank using a solvent system consisting of a mixture of n-butanol, water, and formic acid (7 : 2 : 1) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow the solvent to evaporate in warm circulating air. Spray with Folin-Ciocalteu Phenol TS, followed by sodium carbonate solution (1 in 10): the RF value of the principal spot obtained from the Test solution corresponds to that obtained from the Epinephrine standard solution. Any spot obtained from the Test solution is not larger nor more intense than the spot with the same RF value obtained from the Norepinephrine standard solution, corresponding to not more than 4.0% of norepinephrine.
) coated with a 0.25-mm layer of chromatographic silica gel mixture. Allow to dry, and develop the chromatogram in an unsaturated tank using a solvent system consisting of a mixture of n-butanol, water, and formic acid (7 : 2 : 1) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow the solvent to evaporate in warm circulating air. Spray with Folin-Ciocalteu Phenol TS, followed by sodium carbonate solution (1 in 10): the RF value of the principal spot obtained from the Test solution corresponds to that obtained from the Epinephrine standard solution. Any spot obtained from the Test solution is not larger nor more intense than the spot with the same RF value obtained from the Norepinephrine standard solution, corresponding to not more than 4.0% of norepinephrine.
Assay—
Ferro-citrate solution, Buffer solution, and Standard preparation—
Prepare as directed under Epinephrine Assay  391
391 .
.
Assay preparation—
Transfer about 10 mg of Racepinephrine, accurately weighed, to a 1-liter volumetric flask. Dilute with sodium bisulfite solution (1 in 500) to volume, and mix.
Procedure—
Proceed as directed for Procedure under Epinephrine Assay  391
391 . Calculate the quantity, in mg, of C9H13NO3 in the portion of Racepinephrine taken by the formula:
. Calculate the quantity, in mg, of C9H13NO3 in the portion of Racepinephrine taken by the formula:
(183.20 / 333.29)C(AU / AS)
in which 183.20 and 333.29 are the molecular weights of racepinephrine and epinephrine bitartrate, respectively, C is the concentration, in µg per mL, of USP Epinephrine Bitartrate RS in the Standard preparation, and AU and AS are the absorbances of the solutions from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Kahkashan Zaidi, Ph.D.
Senior Scientist 1-301-816-8269 |
(AER05) Aerosols05 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3469
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.