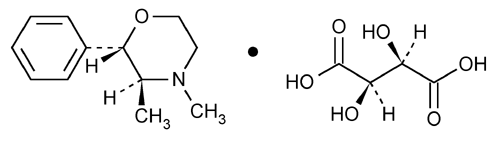
Phendimetrazine Tartrate
Morpholine, 3,4-dimethyl-2-phenyl-, (2S-trans)-, [R-(R*,R*)]-2,3-dihydroxybutanedioate (1:1).
(2S,3S)-3,4-Dimethyl-2-phenylmorpholine l-(+)-tartrate (1:1)
» Phendimetrazine Tartrate contains not less than 98.0 percent and not more than 102.0 percent of C12H17NO·C4H6O6, calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Identification—
C:
It responds to the test for Tartrate  191
191 .
.
Melting range  741
741 :
between 182
:
between 182 and 188
and 188 , with decomposition, but the range between beginning and end of melting does not exceed 3
, with decomposition, but the range between beginning and end of melting does not exceed 3 .
.
pH  791
791 :
between 3.0 and 4.0, in a solution (1 in 40).
:
between 3.0 and 4.0, in a solution (1 in 40).
Loss on drying  731
731 —
Dry it to constant weight at 105
—
Dry it to constant weight at 105 : it loses not more than 0.5% of its weight.
: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Chloride  221
221 —
A 1.0-g portion shows no more chloride than corresponds to 0.50 mL of 0.020 N hydrochloric acid (0.035%).
—
A 1.0-g portion shows no more chloride than corresponds to 0.50 mL of 0.020 N hydrochloric acid (0.035%).
Sulfate  221
221 —
A 1.0-g portion shows no more sulfate than corresponds to 0.10 mL of 0.020 N sulfuric acid (0.01%).
—
A 1.0-g portion shows no more sulfate than corresponds to 0.10 mL of 0.020 N sulfuric acid (0.01%).
Heavy metals  231
231 :
0.001%.
:
0.001%.
Chromatographic purity—
Dissolve 500 mg in water, dilute with water to 5.0 mL, and mix. Apply 10 µL of this preparation and 10 µL of an aqueous solution of USP Phendimetrazine Tartrate RS containing about 100 mg per mL to the starting line to a suitable thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel mixture. Develop the chromatogram in a suitable chamber with a solvent system consisting of a mixture of acetone, methanol, and ammonium hydroxide (50:50:1) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber, air-dry, view under short-wavelength UV light, and observe the location of the spots. Expose the plate to iodine vapors in a closed chamber: yellow spots appear at the same locations as the spots observed under UV light, and the RF value of the spot obtained from the test preparation corresponds to that obtained from the Standard solution, and no other spot is obtained.
) coated with a 0.25-mm layer of chromatographic silica gel mixture. Develop the chromatogram in a suitable chamber with a solvent system consisting of a mixture of acetone, methanol, and ammonium hydroxide (50:50:1) until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber, air-dry, view under short-wavelength UV light, and observe the location of the spots. Expose the plate to iodine vapors in a closed chamber: yellow spots appear at the same locations as the spots observed under UV light, and the RF value of the spot obtained from the test preparation corresponds to that obtained from the Standard solution, and no other spot is obtained.
l-erythro isomer—
Dissolve 3.0 g of Phendimetrazine Tartrate in 25 mL of sodium hydroxide solution (1 in 20) in a suitable separator. Add 25 mL of sodium hydroxide solution (1 in 2), swirl, and allow the phendimetrazine base to separate. Discard the lower, alkaline layer, and collect the upper layer, centrifuging, if necessary, to obtain a clear liquid. Inject 1.0 µL of this liquid into a suitable gas chromatograph equipped with a flame-ionization detector, a 100:1 specimen splitter, and a 25-m × 0.25-mm capillary column, the inside wall of which is coated with a 0.4-µm film of liquid phase G1. The temperatures of the injection port, column, and detector block are 250 , 140
, 140 , and 280
, and 280 , respectively. The carrier gas is helium. Preferably using an electronic integrator, determine the areas of all peaks in the chromatogram. The retention times are about 8.5 minutes for the d-threo isomer and 9 minutes for the l-erythro isomer. Calculate the percentage of l-erythro isomer in the test specimen taken by the formula:
, respectively. The carrier gas is helium. Preferably using an electronic integrator, determine the areas of all peaks in the chromatogram. The retention times are about 8.5 minutes for the d-threo isomer and 9 minutes for the l-erythro isomer. Calculate the percentage of l-erythro isomer in the test specimen taken by the formula:
100(rU / rS)
in which rU is the peak area response of the l-erythro isomer peak and rS is the sum of the areas of the l-erythro isomer peak and the d-threo isomer peak: the limit is 0.1%.
Assay—
Transfer to a beaker about 500 mg of Phendimetrazine Tartrate, accurately weighed, and dissolve in 50 mL of glacial acetic acid. Add 1 drop of crystal violet TS, and titrate with 0.1 N perchloric acid VS to a green endpoint. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 34.14 mg of C12H17NO·C4H6O6.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Clydewyn M. Anthony, Ph.D.
Scientist 1-301-816-8139 |
(MDCCA05) Monograph Development-Cough Cold and Analgesics |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3265
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.