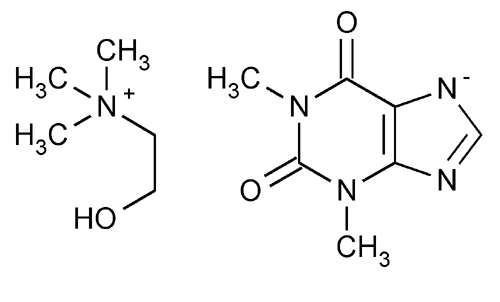
Oxtriphylline
Ethanaminium, 2-hydroxy-N,N,N-trimethyl-, salt with 3,7-dihydro-1,3-dimethyl-1H-purine-2,6-dione.
Choline salt with theophylline (1:1)
» Oxtriphylline contains not less than 61.7 percent and not more than 65.5 percent of anhydrous theophylline (C7H8N4O2), calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Identification—
Dissolve about 1 g in 20 mL of water, and use the solution for the following tests.
A:
To 10 mL of the test solution add 5 mL of mercuric-potassium iodide TS: a pale yellow precipitate is formed (presence of choline).
B:
To 10 mL of the test solution add 5 drops of 6 N ammonium hydroxide and 5 mL of silver nitrate TS: a gelatinous precipitate is formed, and it coagulates on heating (presence of theophylline).
Loss on drying  731
731 —
Dry it at 80
—
Dry it at 80 for 4 hours: it loses not more than 1.0% of its weight.
for 4 hours: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.3%.
:
not more than 0.3%.
Chloride  221
221 —
A 0.50-g portion shows no more chloride than corresponds to 0.15 mL of 0.020 N hydrochloric acid (0.02%).
—
A 0.50-g portion shows no more chloride than corresponds to 0.15 mL of 0.020 N hydrochloric acid (0.02%).
Ordinary impurities  466
466 —
—
Test solution:
a mixture of chloroform, alcohol, and formic acid (88:10:2).
Standard solution:
a mixture of chloroform, alcohol, and formic acid (88:10:2).
Eluant:
a mixture of chloroform, alcohol, and formic acid (88:10:2).
Visualization:
1.
Choline content—
Dissolve about 900 mg of Oxtriphylline, accurately weighed, in 50 mL of water, and add 4 drops of a solution prepared by dissolving 30 mg of methyl red in 100 mL of methanol, adding 15 mL of methylene blue solution (1 in 1000), and mixing. Mix, and titrate with 0.1 N sulfuric acid VS to a purple endpoint. Each mL of 0.1 N sulfuric acid is equivalent to 12.12 mg of C5H15NO2. The content of choline (C5H15NO2) is between 652 mg and 693 mg per g of C7H8N4O2 found in the Assay. Retain the final solution for the Assay for theophylline.
Assay for theophylline—
To the solution retained in the Choline content add 35 mL of silver nitrate TS, swirl gently to promote complete precipitation, and titrate with 0.1 N sodium hydroxide VS to a green endpoint. Each mL of 0.1 N sodium hydroxide is equivalent to 18.02 mg of C7H8N4O2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Daniel K. Bempong, Ph.D.
Senior Scientist 1-301-816-8143 |
(MDPS05) Monograph Development-Pulmonary and Steroids |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3158
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.