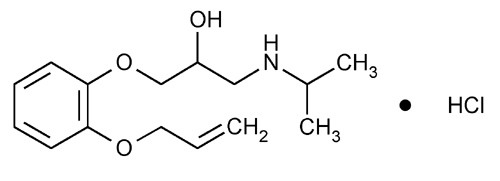
Oxprenolol Hydrochloride
2-Propanol, 1-(o-allyloxyphenoxy)-3-isopropylamino-, hydrochloride.
1-(o-Allyloxyphenoxy)-3-isopropylamino-2-propanol hydrochloride
» Oxprenolol Hydrochloride contains not less than 98.5 percent and not more than 101.0 percent of C15H23NO3·HCl, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
Clarity of solution—
Dissolve 1 g in 10 mL of water: solution is clear.
Identification—
B:
A solution of it responds to the tests for Chloride  191
191 .
.
pH  791
791 :
between 4.0 and 6.0, in a solution (1 in 10).
:
between 4.0 and 6.0, in a solution (1 in 10).
Loss on drying  731
731 —
Dry about 3 g of it in vacuum at 60
—
Dry about 3 g of it in vacuum at 60 for 6 hours: it loses not more than 0.5% of its weight.
for 6 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method II  231
231 :
0.001%.
:
0.001%.
Chromatographic purity—
Diluting solvent—
Prepare a mixture of chloroform and dehydrated alcohol (1:1).
Standard solution A—
Prepare a solution of USP Oxprenolol Hydrochloride RS in Diluting solvent containing 20 mg per mL.
Standard solution B—
Dilute an accurately measured volume of Standard solution A quantitatively, and stepwise if necessary, with Diluting solvent to obtain a solution containing 0.08 mg per mL.
Test solution—
Transfer 200 mg of it to a 10-mL volumetric flask, dissolve in Diluting solvent, dilute with Diluting solvent to volume, and mix.
Procedure—
Apply separate 5-µL portions of the Test solution and the Standard solutions to the starting line of a suitable thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel mixture, previously washed with methanol until the solvent front reaches the top of the plate, dried first in air and then at 100
) coated with a 0.25-mm layer of chromatographic silica gel mixture, previously washed with methanol until the solvent front reaches the top of the plate, dried first in air and then at 100 for 20 minutes, and cooled in a desiccator. Allow the spots to dry. Line a suitable chromatographic chamber with filter paper, saturate the paper with 100 mL of a solvent system consisting of a mixture of ethyl acetate, glacial acetic acid, and water (15:5:5), and allow to stand for about 30 minutes. Place the plate in the chamber, and develop the chromatogram until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber and dry at 100
for 20 minutes, and cooled in a desiccator. Allow the spots to dry. Line a suitable chromatographic chamber with filter paper, saturate the paper with 100 mL of a solvent system consisting of a mixture of ethyl acetate, glacial acetic acid, and water (15:5:5), and allow to stand for about 30 minutes. Place the plate in the chamber, and develop the chromatogram until the solvent front has moved about three-fourths of the length of the plate. Remove the plate from the chamber and dry at 100 for 15 minutes. Spray the plate uniformly with a detection reagent consisting of a freshly prepared mixture of equal volumes of potassium ferricyanide solution (1 in 100) and ferric chloride solution (1 in 5). Dry the plate in a current of warm air for about 5 minutes or until a spot from Standard solution B is visible. Examine the chromatograms in ordinary light: the RF value of the principal spot from the Test solution corresponds to that obtained from Standard solution A. No spot other than the principal spot obtained from the Test solution exceeds in size or intensity the principal spot obtained from Standard solution B (0.4%, corresponding to 0.2% of related compounds, the response factors for which are about double that of oxprenolol hydrochloride).
for 15 minutes. Spray the plate uniformly with a detection reagent consisting of a freshly prepared mixture of equal volumes of potassium ferricyanide solution (1 in 100) and ferric chloride solution (1 in 5). Dry the plate in a current of warm air for about 5 minutes or until a spot from Standard solution B is visible. Examine the chromatograms in ordinary light: the RF value of the principal spot from the Test solution corresponds to that obtained from Standard solution A. No spot other than the principal spot obtained from the Test solution exceeds in size or intensity the principal spot obtained from Standard solution B (0.4%, corresponding to 0.2% of related compounds, the response factors for which are about double that of oxprenolol hydrochloride).
Assay—
Dissolve about 450 mg of Oxprenolol Hydrochloride in 100 mL of glacial acetic acid. Add 10 mL of mercuric acetate TS, and titrate with 0.1 N perchloric acid VS, determining the endpoint potentiometrically, using a glass-calomel electrode system (with a salt bridge of a saturated solution of lithium chloride in glacial acetic acid). Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 30.18 mg of C15H23NO3·HCl.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Sujatha Ramakrishna, Ph.D.
Scientist 1-301-816-8349 |
(MDCV05) Monograph Development-Cardiovascular |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3156
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.