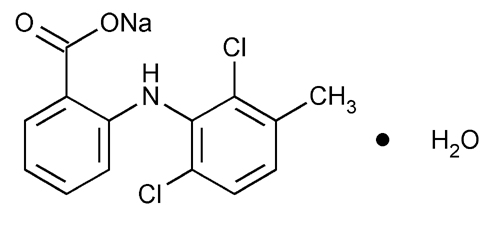
Meclofenamate Sodium
Benzoic acid, 2-[(2,6-dichloro-3-methylphenyl)amino]-, monosodium salt, monohydrate.
Monosodium N-(2,6-dichloro-m-tolyl)anthranilate monohydrate
Anhydrous 318.13
» Meclofenamate Sodium contains not less than 97.0 percent and not more than 103.0 percent of C14H10Cl2NNaO2, calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
B:
Ultraviolet Absorption 1  197U
197U —
—
Solution:
25 µg per mL.
Medium:
0.01 N hydrochloric acid in methanol.
Absorptivities at 242 nm, 279 nm, and 336 nm, calculated on the anhydrous basis, do not differ by more than 3.0%.
C:
Ultraviolet Absorption 2  197U
197U —
—
Solution:
1 in 40,000.
Medium:
0.1 N sodium hydroxide.
Absorptivities at 279 nm and 317 nm, calculated on the anhydrous basis, do not differ by more than 3.0%.
Water, Method I  921
921 :
between 4.8% and 5.8%.
:
between 4.8% and 5.8%.
Copper—
Standard copper solution—
Dissolve 1000 mg of copper wire in 6 mL of nitric acid in a 1 L volumetric flask. Add 8 mL of hydrochloric acid, dilute with water to volume, and mix. Dilute this solution quantitatively and stepwise with water to obtain a Standard copper solution having a known concentration of 0.6 µg per mL.
Test solution—
Transfer 2 g of Meclofenamate Sodium, accurately weighed, to a 100-mL volumetric flask, and add 1 drop of ammonium hydroxide. Dissolve in water, dilute with water to volume, and mix.
Procedure—
Concomitantly determine the absorbances of the Standard copper solution and the Test solution at the copper emission line at about 325 nm, with a suitable atomic absorption spectrophotometer (see Spectrophotometry and Light-scattering  851
851 ) equipped with a copper hollow-cathode lamp, using water as the blank. Adjust the operating conditions to obtain about 70% full-scale detector response with the Standard copper solution. The detector response obtained with the Test solution is not greater than that obtained with the Standard copper solution (0.003%).
) equipped with a copper hollow-cathode lamp, using water as the blank. Adjust the operating conditions to obtain about 70% full-scale detector response with the Standard copper solution. The detector response obtained with the Test solution is not greater than that obtained with the Standard copper solution (0.003%).
Chromatographic purity—
Standard solutions—
Dissolve an accurately weighed quantity of USP Meclofenamate Sodium RS in methanol to obtain a solution containing 20 mg per mL (Standard solution A). Dilute 1.0 mL of Standard solution A with sufficient methanol to obtain 200 mL of solution (Standard solution B).
Test solution—
Dissolve 200 mg of Meclofenamate Sodium in 10.0 mL of methanol.
Procedure—
Apply 10-µL portions of Standard solution A, Standard solution B, and the Test solution to a suitable thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel mixture. Allow the spots to dry, and develop the chromatogram in a solvent system consisting of a mixture of methylene chloride, methyl ethyl ketone, and glacial acetic acid (50:48:2) until the solvent front has moved about eight-tenths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow the solvent to evaporate. Examine the plate under short-wavelength UV light: the chromatograms show a principal spot at about the same RF value, and any secondary spot, if present in the chromatogram from the Test solution is not more intense than the principal spot obtained from Standard solution B (0.5%).
) coated with a 0.25-mm layer of chromatographic silica gel mixture. Allow the spots to dry, and develop the chromatogram in a solvent system consisting of a mixture of methylene chloride, methyl ethyl ketone, and glacial acetic acid (50:48:2) until the solvent front has moved about eight-tenths of the length of the plate. Remove the plate from the developing chamber, mark the solvent front, and allow the solvent to evaporate. Examine the plate under short-wavelength UV light: the chromatograms show a principal spot at about the same RF value, and any secondary spot, if present in the chromatogram from the Test solution is not more intense than the principal spot obtained from Standard solution B (0.5%).
Assay—
Transfer about 350 mg of Meclofenamate Sodium, accurately weighed, to a 125-mL separator, add 10 mL of water, and mix to dissolve. To this solution add 3 mL of 3 N hydrochloric acid, shake, and extract with three 30-mL portions of chloroform, collecting the chloroform extracts in an evaporating flask. Evaporate the chloroform extracts to dryness. Dissolve the residue in 5 mL of dimethyl sulfoxide and 25 mL of methanol. Mix, add 5 drops of phenolphthalein TS, and titrate the mixture with 0.1 N sodium hydroxide VS. Each mL of 0.1 N sodium hydroxide is equivalent to 31.81 mg of C14H10Cl2NNaO2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Clydewyn M. Anthony, Ph.D.
Scientist 1-301-816-8139 |
(MDCCA05) Monograph Development-Cough Cold and Analgesics |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2861
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.