Lindane
Cyclohexane, 1,2,3,4,5,6-hexachloro-, (1
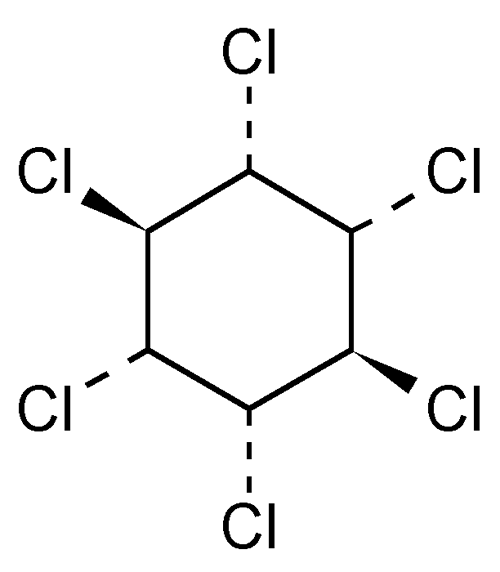
» Lindane is the gamma isomer of hexachlorocyclohexane. It contains not less than 99.0 percent and not more than 101.0 percent of lindane (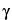 -C6H6Cl6).
-C6H6Cl6).
Packaging and storage—
Preserve in well-closed containers.
Identification, Infrared Absorption  197K
197K .
.
Congealing temperature  651
651 :
not less than 112.0
:
not less than 112.0 .
.
Water, Method I  921
921 :
not more than 0.5%.
:
not more than 0.5%.
Chloride ion—
Place about 100 mg in a test tube with 10 mL of water, shake, and filter. Add 1 mL of nitric acid and 3 mL of silver nitrate TS to the filtrate: no turbidity develops.
Assay—
Internal standard solution—
Dissolve n-octadecane in methylene chloride to obtain a solution having a concentration of about 0.5 mg per mL.
Standard preparation—
Dissolve an accurately weighed quantity of USP Lindane RS in Internal standard solution to obtain a solution having a known concentration of about 2 mg per mL.
System suitability solution—
Prepare solutions of 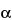 -benzene hexachlorides (BHC) at 1000 µg per mL of methanol,
-benzene hexachlorides (BHC) at 1000 µg per mL of methanol,  -BHC at 1000 µg per mL of acetone, and
-BHC at 1000 µg per mL of acetone, and 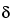 -BHC at 1000 µg per mL of methanol. Transfer 100 µL each of
-BHC at 1000 µg per mL of methanol. Transfer 100 µL each of 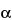 -BHC,
-BHC,  -BHC and
-BHC and 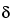 -BHC solutions to a 4-mL conical vial, and evaporate under a stream of nitrogen to dryness. Add to the vial a 100-µL aliquot of the Standard preparation. Insert the stopper, and shake vigorously to dissolve the residue.
-BHC solutions to a 4-mL conical vial, and evaporate under a stream of nitrogen to dryness. Add to the vial a 100-µL aliquot of the Standard preparation. Insert the stopper, and shake vigorously to dissolve the residue.
Assay preparation—
Transfer about 10 mg of Lindane, accurately weighed, to a 5-mL volumetric flask. Dissolve in and dilute with methylene chloride to volume.
Chromatographic system
(see Chromatography  621
621 )—The gas chromatograph is equipped with a flame-ionization detector and a 0.32-mm×30-m fused-silica column coated with a 1-µm phase G46. The chromatograph is programmed as follows. The initial column temperature is maintained at 120
)—The gas chromatograph is equipped with a flame-ionization detector and a 0.32-mm×30-m fused-silica column coated with a 1-µm phase G46. The chromatograph is programmed as follows. The initial column temperature is maintained at 120 for 1 minute. Then, the temperature is increased at a rate of 20
for 1 minute. Then, the temperature is increased at a rate of 20 per minute to 150
per minute to 150 , and then ramped at a rate of 10
, and then ramped at a rate of 10 per minute to 280
per minute to 280 and maintained at that temperature for 4 minutes. The injection port and detector temperatures are maintained at 300
and maintained at that temperature for 4 minutes. The injection port and detector temperatures are maintained at 300 . The injection split ratio is 50:1. Chromatograph the Standard preparation and the System suitability solution, and record the peak responses as directed for Procedure: the relative retention times for n-octadecane and lindane are about 0.85 and 1.0, respectively. [note—Typical retention times for
. The injection split ratio is 50:1. Chromatograph the Standard preparation and the System suitability solution, and record the peak responses as directed for Procedure: the relative retention times for n-octadecane and lindane are about 0.85 and 1.0, respectively. [note—Typical retention times for 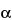 -BHC,
-BHC,  -BHC,
-BHC, 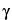 -BHC,
-BHC, 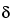 -BHC, and n-octadecane are 15.7, 17.8, 16.5, 18.8, and 13.9 minutes, respectively.] The resolution, R, between n-octadecane and
-BHC, and n-octadecane are 15.7, 17.8, 16.5, 18.8, and 13.9 minutes, respectively.] The resolution, R, between n-octadecane and 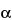 -BHC is not less than 21, between lindane (
-BHC is not less than 21, between lindane (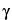 -BHC) and
-BHC) and 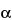 -BHC is not less than 9, between
-BHC is not less than 9, between  -BHC and lindane is not less than 14, and between
-BHC and lindane is not less than 14, and between 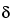 -BHC and
-BHC and  -BHC is not less than 8; the tailing factors for n-octadecane and lindane are less than 1.5 and 1.2, respectively; and the relative standard deviation of the ratios of peak area responses of lindane to n-octadecane for replicate injections of Standard preparation is not more than 1.5%.
-BHC is not less than 8; the tailing factors for n-octadecane and lindane are less than 1.5 and 1.2, respectively; and the relative standard deviation of the ratios of peak area responses of lindane to n-octadecane for replicate injections of Standard preparation is not more than 1.5%.
Procedure—
Separately inject equal volumes (about 1 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of 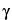 -C6H6Cl6 in the portion of Lindane taken by the formula:
-C6H6Cl6 in the portion of Lindane taken by the formula:
5C(RU / RS)
in which C is the concentration, in mg per mL, of USP Lindane RS in the Standard preparation; and RU and RS are the ratios of the peak responses of lindane to n-octadecane, obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Behnam Davani, Ph.D., M.B.A.
Senior Scientist 1-301-816-8394 |
(MDAA05) Monograph Development-Antivirals and Antimicrobials |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2788
Pharmacopeial Forum: Volume No. 34(2) Page 280
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.