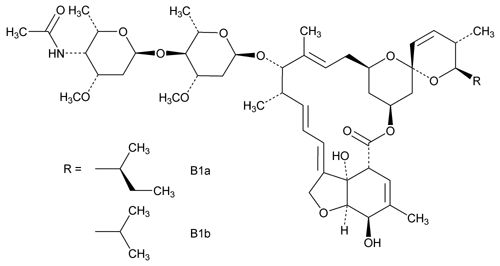
Eprinomectin
C49H73NO14 (Component B1b)




 900.10.
900.10.
Component B1a
Avermectin A1a, 4¢¢-(acetylamino)-5-O-demethyl-4¢¢-deoxy-, (4¢¢R)-.
(2aE,4E,5¢S,6S,6¢R,7S,8E,11R,13S,15S,17aR,20R,20aR,20bS)-6¢-(S)-sec-Butyl-5¢,6,6¢,7,10,11,14,15,17a,20,20a,20b-dodecahydro-20,20b-dihydroxy-5¢,6,8,19-tetramethyl-17-oxospiro[11,15-methano-2H,13H,17H-furo[4,3,2-pq][2,6]benzodioxacyclooctadecin-13,2¢-[2H]pyran]-7-yl-4-O-(4-acetamido-2,4,6-trideoxy-3-O-methyl-
Component B1b
Avermectin A1a, 4¢¢-(acetylamino)-5-O-demethyl-25-de(1-methylpropyl)-4¢¢-deoxy-25-(1-methylethyl)-, (4¢¢R)-.
(2aE,4E,5¢S,6S,6¢R,7S,8E,11R,13S,15S,17aR,20R,20aR,20bS)-5¢,6,6¢,7,10,11,14,15,17a,20,20a,20b-Dodecahydro-20,20b-dihydroxy-6¢-isopropyl-5¢,6,8,19-tetramethyl-17-oxospiro[11,15-methano-2H,13H,17H-furo[4,3,2-pq][2,6]benzodioxacyclooctadecin-13,2¢-[2H]pyran]-7-yl-4-O-(4-acetamido-2,4,6-trideoxy-3-O-methyl-
» Eprinomectin is a mixture of Component B1a and Component B1b. It contains not less than 90.0 percent of Component B1a and not less than 95.0 percent of Components B1a and B1b, calculated on the anhydrous, solvent-free, and antioxidant-free basis. It may contain small amounts of a suitable antioxidant.
Packaging and storage—
Preserve in tight containers, and store between 2 and 8
and 8 at ambient humidity.
at ambient humidity.
Labeling—
Label it to state the name(s) and amount(s) of any added substance(s). Label to indicate that it is for veterinary use only.
Identification—
B:
The retention times of the Component B1a peak and the Component B1b peak in the chromatogram of the Assay preparation correspond to those in the chromatogram of the Standard preparation, as obtained in the Assay.
Specific rotation  781S
781S :
between +132
:
between +132 and +140
and +140 , determined at 405 nm on the anhydrous, solvent-free, and antioxidant-free basis.
, determined at 405 nm on the anhydrous, solvent-free, and antioxidant-free basis.
Test solution:
5 mg per mL, in chloroform.
Water, Method Ia  921
921 :
not more than 2.0%, determined on 0.250 g.
:
not more than 2.0%, determined on 0.250 g.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method II  231
231 :
not more than 10 ppm.
:
not more than 10 ppm.
Limit of residual solvents—
Standard solution I—
Transfer 3.0 mL of acetonitrile, 3.0 mL of methanol, 3.0 mL of isopropyl acetate, and 3.0 mL of heptane to a 50-mL volumetric flask. Dilute with dimethylacetamide to volume, and mix well.
Standard solution II—
Transfer 1.0 mL of Standard solution I to a 100-mL volumetric flask, dilute with dimethylacetamide to volume, and mix well. Further dilute 10.0 mL of this solution with dimethylacetamide to 50.0 mL, and mix well
Test solution—
Transfer 1 g of Eprinomectin in dimethylacetamide to a 10-mL volumetric flask. Dilute with dimethylacetamide to volume, and mix well.
Sensitivity solution I—
Transfer 3.0 mL each of methanol, isopropyl acetate, and heptane to a 50-mL volumetric flask. Dilute with dimethylacetamide to volume, and mix well. Further dilute 50 µL of this solution with dimethylacetamide to 25 mL, and mix well.
Sensitivity solution II—
Transfer 3.0 mL of acetonitrile to a 50-mL volumetric flask, dilute with dimethylacetamide to volume, and mix well. Further dilute 50 µL of this solution with dimethylacetamide to 25 mL.
Sensitivity solution III—
Transfer 5.0 mL of Sensitivity solution I and 1.0 mL of Sensitivity solution II to a 50-mL volumetric flask, dilute with dimethylacetamide to volume, and mix well. [note—This solution contains 100 ppm (m/m) of methanol, isopropyl acetate, and heptane and 20 ppm (m/m) of acetonitrile.]
Chromatographic system (see Chromatography  621
621 )—
The gas chromatograph is equipped with a flame-ionization detector and a 0.53-mm × 25-m fused-silica analytical column coated with a 20-µm S3 stationary phase. The carrier gas is helium with a flow rate of 20 mL per minute. The chromatograph is programmed as follows: the column temperature is increased from 110
)—
The gas chromatograph is equipped with a flame-ionization detector and a 0.53-mm × 25-m fused-silica analytical column coated with a 20-µm S3 stationary phase. The carrier gas is helium with a flow rate of 20 mL per minute. The chromatograph is programmed as follows: the column temperature is increased from 110 at a rate of 5
at a rate of 5 per minute to 160
per minute to 160 and maintained at 160
and maintained at 160 for 5 minutes. The column temperature is then increased at a rate of 30
for 5 minutes. The column temperature is then increased at a rate of 30 per minute to 220
per minute to 220 and maintained at 220
and maintained at 220 for 25 minutes. The injection port temperature is maintained at 200
for 25 minutes. The injection port temperature is maintained at 200 , and the detector temperature is maintained at 220
, and the detector temperature is maintained at 220 . Chromatograph Sensitivity solution III and Standard solution II as directed for Procedure: in the chromatogram obtained from Sensitivity solution III, the peaks for methanol, acetonitrile, isopropyl acetate, and heptane are detectable and elute at relative retention times of 1, 2.1, 7.6, and 8.6, respectively. In the chromatogram obtained from Standard solution II, the relative standard deviations for the areas of the solvent peaks are not more than 5.0% for six injections.
. Chromatograph Sensitivity solution III and Standard solution II as directed for Procedure: in the chromatogram obtained from Sensitivity solution III, the peaks for methanol, acetonitrile, isopropyl acetate, and heptane are detectable and elute at relative retention times of 1, 2.1, 7.6, and 8.6, respectively. In the chromatogram obtained from Standard solution II, the relative standard deviations for the areas of the solvent peaks are not more than 5.0% for six injections.
Procedure—
Separately inject equal volumes (about 1 µL) of Standard solution II and the Test solution. Reinject Standard solution II in duplicate after every six sample injections. The individual values for the area response of the two injections agree within ±5% of their corresponding average response. Calculate the percentage of each solvent present using the following formula:
0.12D(rU / rS)
in which D is the density, in mg per mL, of acetonitrile (0.787), isopropyl acetate (0.870), methanol (0.796), and heptane (0.684); rU is the solvent peak area in the chromatogram obtained from the Test solution; and rS is the solvent peak area in the chromatogram obtained from Standard solution II. Not more than 0.005% of acetonitrile is found, and the sum of all solvents is not more than 0.5%.
Limit of 8a-oxo-B1a—
Solution A, Solution B, Diluent, and System suitability solution—
Proceed as directed in the Assay.
System suitability mobile phase—
Use the Mobile phase as directed in the Assay.
Mobile phase—
Use a mixture of acetonitrile and Solution A (13:7). Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Standard solution—
Use the Standard preparation, prepared as directed in the Assay.
Test solution—
Use the Assay preparation, prepared as directed in the Assay.
Butylated hydroxytoluene solution—
Transfer 50 mg of butylated hydroxytoluene to a 100-mL volumetric flask, and dilute with methanol to volume. Sonicate, if necessary, and mix well. Dilute 2 mL of the resulting solution with Diluent to 100 mL.
System suitability determination—
Use the conditions as directed for Chromatographic system in the Assay.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 280-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L7. The flow rate is 1.5 mL per minute, and the column temperature is 40
)—
The liquid chromatograph is equipped with a 280-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L7. The flow rate is 1.5 mL per minute, and the column temperature is 40 . Chromatograph the Butylated hydroxytoluene solution and the Test solution, and record the peak responses as directed for Procedure: the retention time for the peak corresponding to butylated hydroxytoluene is approximately 12 to 17 minutes, and the relative standard deviation of the peak area is not more than 3.0% for six injections. In the chromatogram obtained from the Test solution, the retention time for the peak corresponding to 8a-oxo-B1a is approximately 4 to 9 minutes.
. Chromatograph the Butylated hydroxytoluene solution and the Test solution, and record the peak responses as directed for Procedure: the retention time for the peak corresponding to butylated hydroxytoluene is approximately 12 to 17 minutes, and the relative standard deviation of the peak area is not more than 3.0% for six injections. In the chromatogram obtained from the Test solution, the retention time for the peak corresponding to 8a-oxo-B1a is approximately 4 to 9 minutes.
Procedure—
Separately inject equal volumes (about 15 µL) of the Butylated hydroxytoluene solution and the Test solution into the chromatograph, record the chromatograms, and measure the areas for the major peaks. Calculate the content of 8a-oxo-B1a, in mg, on the anhydrous, solvent-free, and antioxidant-free basis taken by the formula:
DCPF(rU / rS)
in which D is the dilution factor, in mL, used to prepare the Test solution; C is the concentration, in mg per mL, of butylated hydroxytoluene in the Butylated hydroxytoluene solution; P is the purity of butylated hydroxytoluene used to prepare the Butylated hydroxytoluene solution; F is equal to 0.4 and is the relative response factor for butylated hydroxytoluene with respect to 8a-oxo-B1a; and rU and rS are the peak areas for 8a-oxo-B1a and butylated hydroxytoluene in the chromatograms obtained from the Test solution and the Butylated hydroxytoluene solution, respectively. Not more than 0.5% of 8a-oxo-B1a is found.
Related compounds—
Solution A, Solution B, Mobile phase, Diluent, System suitability solution, and Chromatographic system—
Proceed as directed in the Assay.
Standard solution—
Use the Standard preparation, prepared as directed in the Assay.
Test solution—
Use the Assay preparation, prepared as directed in the Assay.
Procedure—
Inject a volume (about 15 µL) of the Test solution into the chromatograph, record the chromatogram, and measure the peak areas. Calculate the percentage of eprinomectin related compounds in the portion of Eprinomectin taken by the formula:
100(ri / rs)
in which ri is the peak area of each individual related substance obtained from the Test solution, and rs is the sum of the responses of all the peaks: for related compounds with relative retentions of 0.23, 0.93, and 1.16 with respect to the B1a peak, not more than 1.0%; for impurity A, not more than 1.0%; for impurity E, not more than 1.0%; for all other known impurities, not more than 0.5%; for total unknown impurities, not more than 1.0%; and for total impurities, not more than 5.0% is found.
Assay—
Solution A:
0.1% (v/v) solution of perchloric acid in water.
Solution B:
acetonitrile.
Mobile phase—
Use variable mixtures of Solution A and Solution B as directed for Chromatographic system. Make adjustments if necessary (see System Suitability under Chromatography  621
621 ).
).
Diluent—
Prepare a solution of four volumes of methanol and one volume of water.
Standard preparation—
Dissolve an accurately weighed quantity of USP Eprinomectin RS in Diluent to prepare a solution having a known concentration of about 0.500 mg per mL.
System suitability solution—
Transfer 4 mL of Standard preparation to an HPLC vial. Add 2 drops of 1 M sodium hydroxide and let stand for 20 minutes prior to injecting into the chromatograph.
Assay preparation—
Dissolve an accurately weighed quantity of Eprinomectin in Diluent to prepare a solution having a known concentration of about 0.500 mg per mL.
Chromatographic system (see Chromatography  621
621 )—
The liquid chromatograph is equipped with a 245-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L7. The flow rate is 1.5 mL per minute, and the column temperature is 40
)—
The liquid chromatograph is equipped with a 245-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L7. The flow rate is 1.5 mL per minute, and the column temperature is 40 . The chromatograph is programmed as follows:
. The chromatograph is programmed as follows:
Chromatograph the System suitability solution and the Standard preparation as directed for Procedure: the relative retention times are about 0.55, 0.77, 1.00, 1.05, and 1.28 for impurity A, component B1b, component B1a, impurities C + D, and impurity E, respectively; the resolution, R, between components B1b and B1a is not less than 3; the resolution, R, between component B1a and impurities C + D is at least 1; the symmetry factor for the B1a peak is not more than 1.5; and the theoretical plate count for the B1a peak is greater than 4,500. In the chromatogram of the Standard preparation, the relative standard deviation for the peak corresponding to B1a is not more than 1.0% for five injections.
| Time (minute) |
Solution A
(%) |
Solution B
(%) |
|
| 0–15 | 45 | 55 | isocratic |
| 15–25 | 45®5 | 55®95 | linear gradient |
| 25–30 | 5®45 | 95®55 | linear gradient |
| 30–35 | 45 | 55 | isocratic |
Procedure—
Separately inject equal volumes (about 15 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the areas for the major peaks. Calculate the percentage of component B1a by the formula:
100(rU / rT)
in which rU is the peak area of component B1a in the chromatogram obtained from the Assay preparation; and rT is the sum of the peak areas of components B1a and B1b.
Calculate the quantity, in mg, of C50H75NO14 (component B1a) and C49H73NO14 (component B1b) in the portion of Eprinomectin taken by the formula:
DC(rU / rS)
in which D is the dilution factor, in mL, used to prepare the Assay preparation; C is the concentration, in mg per mL, of component B1a or component B1b in the Standard preparation; and rU and rS are the peak areas of component B1a or component B1b in the chromatograms obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Ian DeVeau, Ph.D.
Director, Veterinary Drugs and Radiopharmaceuticals 1-301-816-8178 |
(VET05) Veterinary Drugs 05 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2266
Pharmacopeial Forum: Volume No. 33(1) Page 60
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.