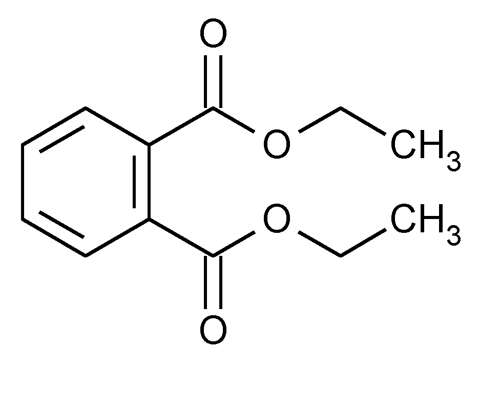
Diethyl Phthalate
» Diethyl Phthalate contains not less than 98.0 percent and not more than 102.0 percent of C12H14O4, calculated on the anhydrous basis.
Caution—Avoid contact.
Packaging and storage—
Preserve in tight containers.
Identification, Infrared Absorption  197F
197F , on undried specimen.
, on undried specimen.
Specific gravity  841
841 :
between 1.118 and 1.122, at 20
:
between 1.118 and 1.122, at 20 .
.
Refractive index  831
831 :
between 1.500 and 1.505, at 20
:
between 1.500 and 1.505, at 20 .
.
Acidity—
Mix 20.0 g with 50 mL of alcohol that previously has been neutralized to a phenolphthalein endpoint, and titrate with 0.10 N sodium hydroxide to a phenolphthalein endpoint: not more than 0.50 mL is required for neutralization.
Water, Method I  921
921 :
not more than 0.2%.
:
not more than 0.2%.
Residue on ignition  281
281 —
Heat gently about 10 g, accurately weighed, until the liquid has evaporated, and ignite the residue to constant weight. Not more than 0.02% is found.
—
Heat gently about 10 g, accurately weighed, until the liquid has evaporated, and ignite the residue to constant weight. Not more than 0.02% is found.
Assay—
Transfer about 1.5 g of Diethyl Phthalate, accurately weighed, to a flask, add 50.0 mL of 0.5 N alcoholic potassium hydroxide VS, attach a reflux condenser to the flask, and boil on a water bath for 1 hour. Add 20 mL of water, then add phenolphthalein TS, and titrate the excess potassium hydroxide with 0.5 N hydrochloric acid VS. Perform a blank determination (see Residual Titrations under Titrimetry  541
541 ). Each mL of 0.5 N potassium hydroxide is equivalent to 55.56 mg of C12H14O4.
). Each mL of 0.5 N potassium hydroxide is equivalent to 55.56 mg of C12H14O4.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Robert H. Lafaver, B.A.
Scientist 1-301-816-8335 |
(EM105) Excipient Monographs 1 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 1226