- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Clemastine Fumarate |
 |
Clemastine Hydrogen Fumarate
(Ph. Eur. monograph 1190)
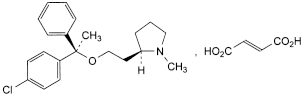
C21H26ClNO,C4H4O4 460.0 14976-57-9
Histamine H1 receptor antagonist; antihistamine.
Ph Eur
(2R)-2-[2-[(R)-1-(4-Chlorophenyl)-1-phenylethoxy]ethyl]-1-methylpyrrolidine (E)-butenedioate.
98.5 per cent to 101.0 per cent (dried substance).
White or almost white, crystalline powder.
Very slightly soluble in water, sparingly soluble in ethanol (70 per cent V/V), slightly soluble in ethanol (50 per cent V/V) and in methanol.
First identification A, B.
Second identification A, C, D.
A. Specific optical rotation (see Tests).
B. Infrared absorption spectrophotometry (2.2.24).
Comparison clemastine fumarate CRS.
C. Examine the chromatograms obtained in the test for related substances.
Results The principal spot in the chromatogram obtained with test solution (b) is similar in position, colour and size to the principal spot in the chromatogram obtained with reference solution (a).
D. Thin-layer chromatography (2.2.27).
Test solution Dissolve 40 mg of the substance to be examined in methanol R and dilute to 2 mL with the same solvent.
Reference solution Dissolve 50 mg of fumaric acid CRS in ethanol (96 per cent) R and dilute to 10 mL with the same solvent.
Plate TLC silica gel G plate R.
Mobile phase water R, anhydrous formic acid R, di-isopropyl ether R (5:25:70 V/V/V).
Application 5 µL.
Development Over a path of 15 cm.
Drying At 100-105 °C for 30 min and allow to cool.
Detection Spray with a 16 g/L solution of potassium permanganate R and examine in daylight.
Results The spot with the highest RF value in the chromatogram obtained with the test solution is similar in position, colour and size to the principal spot in the chromatogram obtained with the reference solution.
Dissolve 0.500 g in methanol R and dilute to 50.0 mL with the same solvent.
Solution S is clear (2.2.1) and not more intensely coloured than reference solution BY7 (2.2.2, Method II).
3.2 to 4.2.
Suspend 1.0 g in 10 mL of carbon dioxide-free water R.
+ 15.0 to + 18.0 (dried substance), determined on solution S.
Thin-layer chromatography (2.2.27).
Test solution (a) Dissolve 0.100 g of the substance to be examined in methanol R and dilute to 5.0 mL with the same solvent.
Test solution (b) Dilute 1.0 mL of test solution (a) to 10.0 mL with methanol R.
Reference solution (a) Dissolve 20.0 mg of clemastine fumarate CRS in methanol R and dilute to 10.0 mL with the same solvent.
Reference solution (b) Dilute 1.5 mL of test solution (b) to 50.0 mL with methanol R.
Reference solution (c) Dilute 0.5 mL of test solution (b) to 50.0 mL with methanol R.
Reference solution (d) Dissolve 10.0 mg of diphenhydramine hydrochloride CRS in 5.0 mL of reference solution (a).
Plate TLC silica gel G plate R.
Mobile phase concentrated ammonia R, methanol R, tetrahydrofuran R (1:20:80 V/V/V).
Application 5 µL.
Development Over a path of 15 cm.
Drying In a current of cold air for 5 min.
Detection Spray with a freshly prepared mixture of 1 volume of potassium iodobismuthate solution R and 10 volumes of dilute acetic acid R and then with dilute hydrogen peroxide solution R; cover the plate immediately with a glass plate of the same size and examine the chromatograms after 2 min.
System suitability Reference solution (d):
- — the chromatogram shows 2 clearly separated spots.
Limits Test solution (a):
- — any impurity: any spot, apart from the principal spot, is not more intense than the principal spot in the chromatogram obtained with reference solution (b) (0.3 per cent) and at most 4 such spots are more intense than the principal spot in the chromatogram obtained with reference solution (c) (0.1 per cent);
- — disregard limit: disregard any spot remaining at the point of application (fumaric acid).
Liquid chromatography (2.2.29).
Solvent mixture acetonitrile R1, 10 g/L solution of ammonium dihydrogen phosphate R (25:75 V/V).
Test solution Dissolve 20 mg of the substance to be examined in the solvent mixture and dilute to 100 mL with the solvent mixture.
Reference solution (a) Dissolve 6 mg of 1-(4-chlorophenyl)-1-phenylethanol CRS (impurity C) in the solvent mixture and dilute to 100 mL with the solvent mixture.
Reference solution (b) Dilute 1 mL of reference solution (a) to 100 mL with the solvent mixture.
Reference solution (c) Dissolve 10 mg of the substance to be examined in the solvent mixture and dilute to 100 mL with the solvent mixture. To 1 mL of this solution add 1 mL of reference solution (a) and dilute to 100 mL with the solvent mixture.
- — size: l = 0.1 m, Ø = 4.6 mm;
- — stationary phase: octadecylsilyl silica gel for chromatography R (5 µm).
Mobile phase phosphoric acid R, acetonitrile R1, 10 g/L solution of ammonium dihydrogen phosphate R (0.1:45:55 V/V/V).
Flow rate 1 mL/min.
Detection Spectrophotometer at 220 nm.
Injection 100 µL.
System suitability Reference solution (c):
- — resolution: minimum 2.2 between the peaks due to clemastine and impurity C.
- — impurity C: not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.3 per cent).
Maximum 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C for 6 h.
Maximum 0.1 per cent, determined on 1.0 g.
Dissolve 0.350 g in 60 mL of anhydrous acetic acid R. Titrate with 0.1 M perchloric acid, determining the end-point potentiometrically (2.2.20).
1 mL of 0.1 M perchloric acid is equivalent to 46.00 mg of C25H30ClNO5.
Specified impurities A, B, C.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use): D.
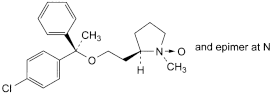
A. (1RS,2R)-2-[2-[(R)-1-(4-chlorophenyl)-1-phenylethoxy]ethyl]-1-methylpyrrolidine 1-oxide,
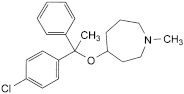
B. 4-[1-(4-chlorophenyl)-1-phenylethoxy]-1-methylazepane,
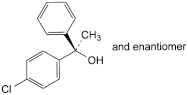
C. (RS)-1-(4-chlorophenyl)-1-phenylethanol,
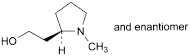
D. 2-[(2RS)-1-methylpyrrolidin-2-yl]ethanol.
Ph Eur

