- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Amantadine Hydrochloride |
 |
(Ph Eur monograph 0463)
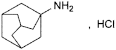
C10H17N,HCl 187.7 665-66-7
Viral replication inhibitor (influenza A); dopamine receptor agonist; treatment of influenza and Parkinson's disease.
Ph Eur
Tricyclo[3.3.1.13,7]decan-1-amine hydrochloride.
98.5 per cent to 101.0 per cent (anhydrous substance).
White or almost white, crystalline powder.
Freely soluble in water and in ethanol (96 per cent).
It sublimes on heating.
First identification A, D.
Second identification B, C, D.
A. Infrared absorption spectrophotometry (2.2.24).
Preparation Discs.
Comparison amantadine hydrochloride CRS.
B. To 0.1 g add 1 mL of pyridine R, mix and add 0.1 mL of acetic anhydride R. Heat to boiling for about 10 s. Pour the hot solution into 10 mL of dilute hydrochloric acid R, cool to 5 °C and filter. The precipitate, washed with water R and dried in vacuo at 60 °C for 1 h, melts (2.2.14) at 147 °C to 151 °C.
C. Dissolve 0.2 g in 1 mL of 0.1 M hydrochloric acid. Add 1 mL of a 500 g/L solution of sodium nitrite R. A white precipitate is formed.
D. 1 mL of solution S (see Tests) gives reaction (a) of chlorides (2.3.1).
Dissolve 2.5 g in carbon dioxide-free water R and dilute to 25 mL with the same solvent.
Solution S is clear (2.2.1) and not more intensely coloured than reference solution Y7 (2.2.2, Method II).
Dilute 2 mL of solution S to 10 mL with carbon dioxide-free water R. Add 0.1 mL of methyl red solution R and 0.2 mL of 0.01 M sodium hydroxide. The solution is yellow. Add 0.4 mL of 0.01 M hydrochloric acid. The solution is red.
Gas chromatography (2.2.28): use the normalisation procedure.
Test solution Dissolve 0.10 g of the substance to be examined in 2 mL of water R. Add 2 mL of a 200 g/L solution of sodium hydroxide R and 2 mL of chloroform R. Shake for 10 min. Separate the chloroform layer, dry over anhydrous sodium sulfate R and filter.
- — material: glass;
- — size: l = 1.8 m, Ø = 2 mm;
- — stationary phase: mix 19.5 g of silanised diatomaceous earth for gas chromatography R with 60 mL of a 3.3 g/L solution of potassium hydroxide R in methanol R and evaporate the solvent under reduced pressure while rotating the mixture slowly (support); dissolve 0.4 g of low-vapour-pressure hydrocarbons (type L) R in 60 mL of toluene R (dissolution requires up to 5 h), add this solution to the support and evaporate the solvent under reduced pressure while rotating the mixture slowly.
Carrier gas nitrogen for chromatography R.
Flow rate 30 mL/min.
Temperature:

Detection Flame ionisation.
Injection 1 µL or the chosen volume.
Run time At least 2.5 times the retention time of amantadine.
- — any impurity: for each impurity, maximum 0.3 per cent;
- — total: maximum 1 per cent;
- — disregard limit: disregard the peak due to the solvent.
Maximum 20 ppm.
12 mL of solution S complies with test A. Prepare the reference solution using lead standard solution (2 ppm Pb) R.
Maximum 0.5 per cent, determined on 2.000 g.
Maximum 0.1 per cent, determined on 1.0 g.
Dissolve 0.150 g in a mixture of 5.0 mL of 0.01 M hydrochloric acid and 50 mL of ethanol (96 per cent) R. Carry out a potentiometric titration (2.2.20), using 0.1 M sodium hydroxide. Read the volume added between the 2 points of inflexion.
1 mL of 0.1 M sodium hydroxide is equivalent to 18.77 mg of C10H18ClN.
Ph Eur