- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Montelukast Sodium |
 |
(Ph. Eur. monograph 2583)
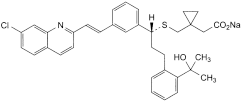
C35H35CINNaO3S 608.0 151767-02-1
Leukotriene CysLT1 receptor antagonist; treatment of asthma.
Ph Eur
Sodium [1-[[[(1R)-1-[3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetate.
98.0 per cent to 102.0 per cent (anhydrous substance).
White or almost white, hygroscopic powder.
Freely soluble in water and in methylene chloride, freely soluble to very soluble in ethanol (96 per cent).
A. Infrared absorption spectrophotometry (2.2.24).
Comparison montelukast sodium CRS.
B. Enantiomeric purity (see Tests).
C. Ignite 0.1 g in a crucible until an almost white residue is obtained. Take up the residue in 2 mL of water R and filter. The filtrate gives reaction (a) of sodium (2.3.1).
Liquid chromatography (2.2.29). Carry out the test protected from light. Prepare the solutions in amber flasks.
Solvent mixture acetonitrile R, water R (50:50 V/V).
Test solution Dissolve 50 mg of the substance to be examined in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Reference solution (a) Dilute 1.0 mL of the test solution to 100.0 mL with the solvent mixture. Dilute 1.0 mL of this solution to 10.0 mL with the solvent mixture.
Reference solution (b) Dissolve 5 mg of montelukast racemate CRS in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
- — size: l = 0.15 m, Ø = 4.0 mm;
- — stationary phase: silica gel AGP for chiral chromatography R (5 µm);
- — temperature: 30 °C.
- — mobile phase A: 2.3 g/L solution of ammonium acetate R adjusted to pH 5.7 with glacial acetic acid R;
- — mobile phase B: acetonitrile R, methanol R (40:60 V/V);

Flow rate 0.9 mL/min.
Detection Spectrophotometer at 280 nm.
Injection 10 µL.
Relative retention With reference to montelukast (retention time = about 25 min): impurity A = about 0.7.
- — resolution: minimum 2.9 between the peaks due to impurity A and montelukast in the chromatogram obtained with reference solution (b);
- — signal-to-noise ratio: minimum 10 for the principal peak in the chromatogram obtained with reference solution (a).
Calculate the percentage content of impurity A using the following expression:
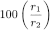
r1 |
= |
area of the peak due to impurity A in the chromatogram obtained with the test solution; |
r2 |
= |
sum of the areas of the peaks due to montelukast and impurity A in the chromatogram obtained with the test solution. |
- — impurity A: maximum 0.2 per cent.
Liquid chromatography (2.2.29): use the normalisation procedure. Carry out the test protected from light. Prepare the solutions in amber flasks.
Solvent mixture water R, methanol R (10:90 V/V).
Test solution (a) Dissolve 50.0 mg of the substance to be examined in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Test solution (b) Dilute 10.0 mL of test solution (a) to 100.0 mL with the solvent mixture.
Reference solution (a) Dilute 1.0 mL of test solution (a) to 100.0 mL with the solvent mixture. Dilute 1.0 mL of this solution to 20.0 mL with the solvent mixture.
Reference solution (b) Dissolve 10 mg of montelukast for peak identification CRS (containing impurities B, C, D, E and F) in the solvent mixture and dilute to 10.0 mL with the solvent mixture.
Reference solution (c) In order to prepare impurity G in situ, transfer 1 mL of reference solution (b) to a colourless glass vial and expose to ambient light for about 20 min.
Reference solution (d) Dissolve 65.0 mg of montelukast dicyclohexylamine CRS in the solvent mixture and dilute to 50.0 mL with the solvent mixture. Dilute 10.0 mL of the solution to 100.0 mL with the solvent mixture.
- — size: l = 0.05 m, Ø = 4.6 mm;
- — stationary phase: phenylsilyl silica gel for chromatography R (1.8 µm);
- — temperature: 30 °C.
- — mobile phase A: mix 1.5 mL of trifluoroacetic acid R and 1000 mL of water R;
- — mobile phase B: mix 1.5 mL of trifluoroacetic acid R and 1000 mL of acetonitrile R;
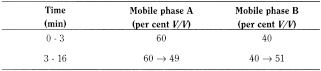
Flow rate 1.2 mL/min.
Detection Spectrophotometer at 238 nm.
Injection 10 µL of test solution (a) and reference solutions (a) and (c); 20 µL of reference solution (b).
Identification of impurities Use the chromatogram supplied with montelukast for peak identification CRS and the chromatogram obtained with reference solution (b) to identify the peaks due to impurities B, C, D, E and F; use the chromatogram obtained with reference solution (c) to identify the peak due to impurity G.
Relative retention With reference to montelukast (retention time = about 7 min): impurity C = about 0.4; impurity G = about 0.8; impurities D and E = about 0.9; impurity F = about 1.2; impurity B = about 1.9.
System suitability Reference solution (c):
- — resolution: minimum 2.5 between the peaks due to impurity G and montelukast; minimum 1.5 between the peaks due to montelukast and impurity F.
- — impurity B: maximum 0.3 per cent;
- — impurity C: maximum 0.2 per cent;
- — impurities F, G: for each impurity, maximum 0.15 per cent;
- — sum of impurities D and E: maximum 0.15 per cent;
- — unspecified impurities: for each impurity, maximum 0.10 per cent;
- — total: maximum 0.6 per cent;
- — disregard limit: the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent).
Maximum 10 ppm.
Solvent mixture water R, acetone R (20:80 V/V).
0.50 g complies with test H. Prepare the reference solution using 0.5 mL of lead standard solution (10 ppm Pb) R.
Maximum 4.0 per cent, determined on 0.300 g.
Liquid chromatography (2.2.29) as described in the test for related substances with the following modifications.
Injection Test solution (b) and reference solution (d).
Calculate the percentage content of C35H35ClNNaO3S using the following expression:
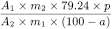
A1 |
= |
area of the principal peak in the chromatogram obtained with test solution (b); |
A2 |
= |
area of the principal peak in the chromatogram obtained with reference solution (d); |
m1 |
= |
mass of the substance to be examined used to prepare test solution (a), in milligrams; |
m2 |
= |
mass of montelukast dicyclohexylamine CRS used to prepare reference solution (d), in milligrams; |
p |
= |
declared percentage content of montelukast dicyclohexylamine CRS; |
a |
= |
percentage content of water in the substance to be examined. |
In an airtight container, protected from light.
Specified impurities A, B, C, D, E, F, G.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use): H, I.
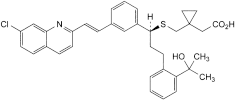
A. [1-[[[(1S)-1-[3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid,
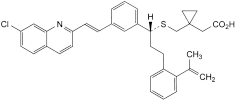
B. [1-[[[(1R)-1-[3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(1-methylethenyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid,
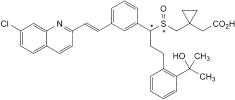
C. [1-[[[1-[3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]sulfinyl]methyl]cyclopropyl]acetic acid,
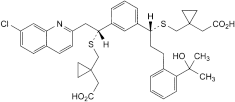
D. 1-[[[(1R)-1-[3-[(1R)-1-[[[1-(carboxymethyl)cyclopropyl]methyl]sulfanyl]-2-(7-chloroquinolin-2-yl)ethyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid,
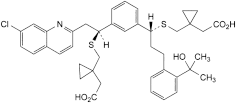
E. 1-[[[(1R)-1-[3-[(1S)-1-[[[1-(carboxymethyl)cyclopropyl]methyl]sulfanyl]-2-(7-chloroquinolin-2-yl)ethyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid,
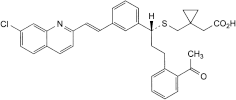
F. [1-[[[(1R)-3-(2-acetylphenyl)-1-[3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid,
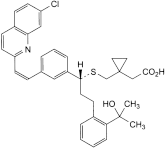
G. [1-[[[(1R)-1-[3-[(Z)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid,
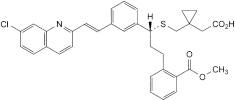
H. [1-[[[(1R)-1-[3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(methoxycarbonyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid,
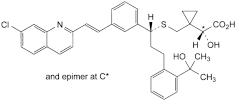
I. (2RS)-[1-[[[(1R)-1-[3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl](hydroxy)acetic acid.
Ph Eur